THERAPY
TAMOXIFEN (INCLUDING NATURAL ALTERNATIVES), AND AROMATASE INHIBITORS (AIs)
"For women with estrogen receptor-positive disease, continuing tamoxifen to 10 years rather than stopping at 5 years produces a further reduction in recurrence and mortality, particularly after year 10. These results, taken together with results from previous trials of 5 years of tamoxifen treatment versus none, suggest that 10 years of tamoxifen treatment can approximately halve breast cancer mortality during the second decade after diagnosis." Davies C et al., 2013.
"As time passed, tamoxifen proved to be the anti-cancer drug that actually caused cancer. As a drug chemist, I was prompted to fix Big Pharma's dirty secret. For one year, I attempted to offset the cancer side effect of tamoxifen by designing chemical cousins of it. Such attempts failed. All of its molecular relatives initiated cancer. The project was canceled. The secret was kept. Tamoxifen remained on the market and continued its cancer scourge among unsuspecting victims."
Shane Ellison, 2007. http://www.newswithviews.com/Ellison/shane30.htm.
"As time passed, tamoxifen proved to be the anti-cancer drug that actually caused cancer. As a drug chemist, I was prompted to fix Big Pharma's dirty secret. For one year, I attempted to offset the cancer side effect of tamoxifen by designing chemical cousins of it. Such attempts failed. All of its molecular relatives initiated cancer. The project was canceled. The secret was kept. Tamoxifen remained on the market and continued its cancer scourge among unsuspecting victims."
Shane Ellison, 2007. http://www.newswithviews.com/Ellison/shane30.htm.
TAMOXIFEN USED IN LIEU OF CHEMO
IN EARLY BREAST CANCER -
VERY LOW RATES OF RECURRENCE IN 2015 PROSPECTIVE STUDY
"Among patients with hormone-receptor–positive, HER2-negative, axillary node–negative breast cancer who met established guidelines for the recommendation of adjuvant chemotherapy on the basis of clinicopathologic features, those with tumors that had a favorable gene-expression profile had very low rates of recurrence at 5 years with endocrine therapy (tamoxifen) alone."
Sparano J et al., Prospective Validation of a 21-Gene Expression Assay in Breast Cancer, NEJM, 2015. SEE MORE IN THE THERAPY-CHEMOTHERAPY SECTION.
IN EARLY BREAST CANCER -
VERY LOW RATES OF RECURRENCE IN 2015 PROSPECTIVE STUDY
"Among patients with hormone-receptor–positive, HER2-negative, axillary node–negative breast cancer who met established guidelines for the recommendation of adjuvant chemotherapy on the basis of clinicopathologic features, those with tumors that had a favorable gene-expression profile had very low rates of recurrence at 5 years with endocrine therapy (tamoxifen) alone."
Sparano J et al., Prospective Validation of a 21-Gene Expression Assay in Breast Cancer, NEJM, 2015. SEE MORE IN THE THERAPY-CHEMOTHERAPY SECTION.
*...Tamoxifen is the
first choice
for premenopausal women
(hormone- positive),
and is still a good choice for postmenopausal women
who can't take
an aromatase inhibitor." *
http://www.breastcancer.org/treatment/hormonal/serms/tamoxifen
first choice
for premenopausal women
(hormone- positive),
and is still a good choice for postmenopausal women
who can't take
an aromatase inhibitor." *
http://www.breastcancer.org/treatment/hormonal/serms/tamoxifen
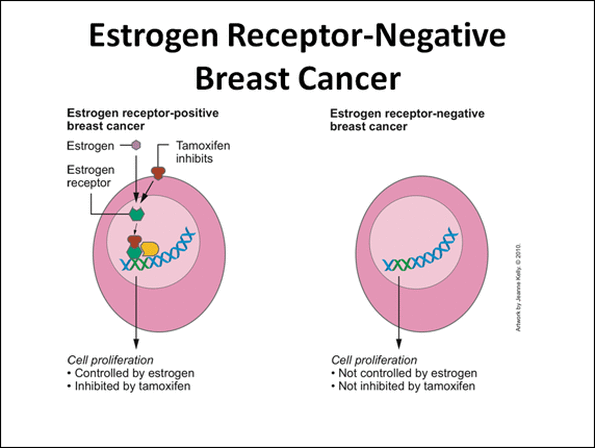
TAMOXIFEN IS NOT FOR ESTROGEN-NEGATIVE BREAST CANCER.
ONLY ESTROGEN-POSITIVE
BREAST CANCER PATIENTS ARE CANDIDATES FOR TAMOXIFEN.
ONLY ESTROGEN-POSITIVE
BREAST CANCER PATIENTS ARE CANDIDATES FOR TAMOXIFEN.
WHAT IS TAMOXIFEN?
TAMOXIFEN IS A SELECTIVE ESTROGEN RECEPTOR MODULATOR (SERM)
From the National Cancer Institute:
"Blocking estrogen’s effects: Several types of drugs interfere with estrogen’s ability to stimulate the growth of breast cancer cells:
- Selective estrogen receptor modulators (SERMs) bind to estrogen receptors, preventing estrogen from binding. Examples of SERMSs approved by the FDA are tamoxifen (Nolvadex®), raloxifene (Evista®), and toremifene (Fareston®). Tamoxifen has been used for more than 30 years to treat hormone receptor-positive breast cancer.
Because SERMs bind to estrogen receptors, they can potentially not only block estrogen activity (i.e., serve as estrogen antagonists) but also mimic estrogen effects (i.e., serve as estrogen agonists). Most SERMs behave as estrogen antagonists in some tissues and as estrogen agonists in other tissues. For example, tamoxifen blocks the effects of estrogen in breast tissue, but acts like estrogen in the uterus and bone." (See http://www.cancer.gov/types/breast/breast-hormone-therapy-fact-sheet.)
-------------------------------
"For example, tamoxifen blocks the effects of estrogen in breast tissue, but acts like estrogen in the uterus and bone." (See citation above.)
"For example, tamoxifen blocks the effects of estrogen in breast tissue, but acts like estrogen in the uterus and bone." (See citation above.)
-------------------------------------------
SUCCESS OF TAMOXIFEN
EXTENDING TREATMENT FROM FIVE YEARS TO TEN YEARS - RESULTS SUGGEST THAT TEN YEARS OF TAMOXIFEN CAN APPROXIMATELY HALVE BREAST CANCER MORTALITY DURING SECOND DECADE AFTER DIAGNOSIS (ATLAS TRIAL)
In the Davies C et al. 2013 study, the researchers investigated whether or not it would be beneficial to extend tamoxifen therapy from five years to ten years for patients who have estrogen receptor-positive, early breast cancer.
"In the worldwide Adjuvant Tamoxifen: Longer Against Shorter (ATLAS) trial, 12 894 women with early breast cancer who had completed 5 years of treatment with tamoxifen were randomly allocated to continue tamoxifen to 10 years or stop at 5 years."
Findings:
The risk of recurrence during years 5–14 was 21·4% for women continuing on tamoxifen for ten years versus 25·1% for women on tamoxifen for five years.
"Breast cancer mortality during years 5–14 was 12·2% for women allocated to continue with tamoxifen versus 15·0% for controls (absolute mortality reduction 2·8%)."
Incidence of hospitalization or death: "Pulmonary embolus 1·87 [including 0·2% mortality in both treatment groups]), stroke 1·06 , ischaemic heart disease , and endometrial cancer 1·74. The cumulative risk of endometrial cancer during years 5–14 was 3·1% (mortality 0·4%) for women allocated to continue versus 1·6% (mortality 0·2%) for controls (absolute mortality increase 0·2%)."
Interpretation: "For women with estrogen receptor-positive disease, continuing tamoxifen to 10 years rather than stopping at 5 years produces a further reduction in recurrence and mortality, particularly after year 10. These results, taken together with results from previous trials of 5 years of tamoxifen treatment versus none, suggest that 10 years of tamoxifen treatment can approximately halve breast cancer mortality during the second decade after diagnosis." (See Davies C et al., Long-Term Effects of Continuing Adjuvant Tamoxifen to 10 Years Versus Stopping at 5 Years After Diagnosis of Estrogen Receptor-Positive Breast Cancer: ATLAS, A Randomized Trial, Lancet Oncology, 2013.)
COMMENT ON ATLAS TAMOXIFEN TRIAL
"Concern about life-threatening adverse events such as thromboembolic events or secondary endometrial cancers has also kept patients from initiating and/or completing adjuvant tamoxifen therapy. If the absolute benefit in terms of decrease in breast cancer–related mortality of an additional 5 years of tamoxifen therapy is only 2.8%, a balanced discussion needs to occur weighing the benefits of therapy vs. the impact on quality of life and potential life-threatening toxicities." (See Smith, G, Long and Short of Tamoxifen Therapy: A Review of the Atlas Trial, J Adv Pract Oncol. 2014.)
WHY TAMOXIFEN MAY BE HELPFUL
TAMOXIFEN AND PRIONS - PRIONS IN BREAST TUMORS? CAN TAMOXIFEN ERADICATE PRIONS IN TUMORS? BLOCKING ESTROGEN RECEPTORS THAT MAINTAIN PRIONS - KEY TO TAMOXIFEN'S SUCCESS IN BREAST CANCER TUMORS?
Perhaps one of the most compelling ways that tamoxifen can be helpful in breast cancer is in its targeting of prions in tumors.
Background: Prions are infectious misfolded proteins. The Rangel L et al. 2014 study stated, "The tumor suppressor protein p53 loses its function in more than 50% of human malignant tumors." This tumor suppressor p53 can become transformed into a mutant p53 and "aggregation of mutant p53 (amyloid ogliomers and fibrils) confers a prion-like activity... The detection of amyloid aggregates in biopsied breast cancer tissues (especially in aggressive forms) demonstrates the relevance of this prionoid behavior in cancer pathogenesis."
Is there any way to eradicate these prion-like fibrillar amyloids?
Use tamoxifen?
In the Molloy B et al. 2014 study, the researchers did not explore the use of tamoxifen to eradicate prions in tumors but, instead, to use tamoxifen to target prions in neurological diseases.
According to the researchers, "...Estrogen acts as a regulator of the processes of both prion infection and prion maintenance. Estrogen was found to be cell biased in its effect; it protected cells against prion infection in a prevention mode and enabled prions in a treatment mode. These processes were regulated by the estrogen receptor subtypes Erα (estrogen receptor a) and Erβ (estrogen receptor b). By using specific receptor agonists, Erα was found to be the main receptor active in slowing prion infection, whereas in chronically infected cells, although Erα allowed partial maintenance of prion levels, Erβ was the main receptor involved in maintaining prions in a treatment paradigm."
Thus, "It has emerged that estrogen's action is affected by neurological health: estrogen has a positive protective effect in healthy cells, whereas it exacerbates conditions in diseased, unhealthy, or aged cells."
Conclusion: And here is where tamoxifen comes into play.
" ... By targeting the estrogen receptors with the selective estrogen receptor modulators tamoxifen and 4-hydroxy-tamoxifen, prions could be cleared from prion-infected cell culture. Tamoxifen and 4-hydroxy-tamoxifen had half-maximal inhibitory concentrations for clearance of prions of 0.47 μM and 0.14 nM, respectively. This work identifies further factors involved in the prion disease process, and through antagonism of the estrogen system, we demonstrate that the estrogen system is a target for controlling prion levels." ... Note that, "Estrogen therapy has been shown to lower the risk of development of Alzheimer's disease; however, in aged rat models, estrogen treatment postneurodegeneration worsened the degeneration process. With this in mind, estrogen application post-prion infection, particularly in an aged rat model, may have a negative impact on the disease process. This is a significant consideration for late-onset prion diseases, especially if the response of the human estrogen receptors mirrors that seen in aged rat models, in which only ERb remains responsive to estrogen." (For the study on prions and breast cancer, see, Rangel L et al., The Aggregation of Mutant p53 Produces Prion -Like Properties in Cancer , Prions, 2014. .... For the study on tamoxifen, estrogen, and neurological diseases, see Molloy B et al., A Cell-Biased Effect of Estrogen in Prion Infection, Journal of Virology, 2014.)
EDITORS' NOTE: Can we extrapolate from the estrogen cell bias in neurological diseases to breast cancer? How about taking estrogen, especially in an older person with breast cancer? If the estrogen receptor, ERb, is basically only active in older breast cancer patients, will this receptor help to maintain the breast cancer tumor harboring these infectious prions? Should older breast cancer patients avoid taking estrogen altogether? How about younger breast cancer patients?
Thus, does tamoxifen play a major, compelling role in blocking the estrogen receptors to clear the prions in breast cancer tumors? A big reason for tamoxifen's reputed success?
Thus, does tamoxifen play a major, compelling role in blocking the estrogen receptors to clear the prions in breast cancer tumors? A big reason for tamoxifen's reputed success?
WHY TAMOXIFEN MAY BE HELPFUL
AS AN ANTIFUNGAL AGAINST SACCHAROMYCES CEREVISIAE, CANDIDA ALBICANS IN BREAST CANCER PATIENTS
Tamoxifen has been found to have antifungal activity against the strain of fungus, Saccharomyces cerevisiae (Wiseman H et al., 1990). And there have been investigations of tamoxifen's effect against Candida albicans.
Background: "Candida albicans is an opportunistic pathogen of humans with an increasing medical relevance, causing superficial as well as systemic infections in susceptible individuals. The secreted aspartyl proteinases (Saps) have been recognized as a virulence-associated trait of the C, albicans pathogen."
In the Wang Y et al. 2013 study, the researchers "investigated the connection between serum Sap2 and different kinds of cancer. In this study, 22 (27.3%) patients with cervical carcinoma were positive for serum Sap2, six (25%) patients with intestinal cancer, and 15 (22.3%) patients with breast cancer were positive for Sap2, while only one (5.9%) patient with hepatocarcinoma was positive for Sap2."
Now that we know, from at least one study, that 22.3% of breast cancer patients harbor candida, will tamoxifen be expected to exert any salutary antifungal activity?
In the Dolan K et al. 2006 study, the researchers "report that tamoxifen displays in vitro activity (MIC, 8 to 64 microg/ml) against pathogenic yeasts (Candida albicans, other Candida spp., and Cryptococcus neoformans). In vivo, 200 mg/kg of body weight per day of tamoxifen reduced kidney fungal burden ... in a murine model of disseminated candidiasis."
How does tamoxifen work against fungus?
In the Lam HY et al 1984 study, the researchers "presented evidence that tamoxifen is an antagonist of calmodulin, a major cellular calcium receptor and calcium dependent regulator of many cellular processes ... It could be speculated that the antagonism of calmodulin by tamoxifen may be one of the mechanisms responsible for its pharmacological actions."
And here is how tamoxifen works as a calmodulin inhibitor in yeast.
From the Dolan group, " ... Tamoxifen-treated yeast show phenotypes consistent with decreased calmodulin function, including lysis, decreased new bud formation, disrupted actin polarization, and decreased germ tube formation." EDITORS' NOTE: Good for breast cancer! (For the candida and cancer study, see Wang Y et al., Detection of Candida Albicans Sap2 in Cancer Patient Serum Samples by an Indirect Competitive Enyzme-Linked Immunosorbent Assay for the Diagnosis of Candidiasis, Indian Journal of Microbiology and Pathology, 2013 ...For the calmodulin mechanism study, see Lam HY et al., Tamoxifen is a Calmodulin Antagonist in the Activation of cAMP Phosphodiesterase, Biochemi Biphys Res Commun, 1984 For the antifungal tamoxifen study, see Dolan K et al., Antifungal Activity of Tamoxifen: In Vitro and In Vivo Activities and Mechanistic Characterization, Antimicrob Agents Chemother, 2009.)
WHY TAMOXIFEN MAY BE HELPFUL
TAMOXIFEN AGAINST LEISHMANIA - IN ADDITION TO BEING AN ESTROGEN RECEPTOR MODULATOR,
OTHER CLUES AS TO HOW TAMOXIFEN MAY BE EFFECTIVE AGAINST BREAST CANCER?
In the past, American service people serving in the Middle East have been given tamoxifen to kill leishmania. "Leishmaniasis is a complex of vector-borne infectious diseases transmitted by sand flies and caused by protozoan parasites of the genus Leishmania."
It is unclear how this SERM, tamoxifen, is able to kill Leishmania, since researchers have not been able to find any "estrogen receptor encoding homologue in the genome of Leishmania."
We know from the Miguel DC et al. 2007 study, that by alkinalizing the cell, going from pH 4.5 to pH 7.5, tamoxifen could more "effectively kill several Leishmania species".
In the 2015 Coelho A et al. study, the researchers stated, "We have previously reported that tamoxifen, a selective estrogen receptor modulator, is active in vitro and in vivo against Leishmania species. We have demonstrated that tamoxifen's antileishmanial activity is not dependent on the interaction with estrogen receptors, but the precise antileishmanial mechanism of action is still uncertain ..... Tamoxifen must have multiple targets in the parasite in agreement with observations in tumor cells where, besides being able to bind and modulate estrogen receptor activity, tamoxifen has been shown to exert other effects such as interference in membranes, ceramide metabolism, calcium-calmodulin modulation and protein kinase C activity."
(See Miguel DC et al., Tamoxifen is Effective Against Leishmania and Induces a Rapid Alkalinization of Parasitophorous Vacuoles Harboring Leishmania Amazonensis Amastigotes, J Antimicrob Chemother, 2007 .... See Coelho A et al., Leishmania is Not Prone to Develop Resistance to Tamoxifen, International Journal for Parasitology, Drugs, and Drug Resistance, 2015.)
In the past, American service people serving in the Middle East have been given tamoxifen to kill leishmania. "Leishmaniasis is a complex of vector-borne infectious diseases transmitted by sand flies and caused by protozoan parasites of the genus Leishmania."
It is unclear how this SERM, tamoxifen, is able to kill Leishmania, since researchers have not been able to find any "estrogen receptor encoding homologue in the genome of Leishmania."
We know from the Miguel DC et al. 2007 study, that by alkinalizing the cell, going from pH 4.5 to pH 7.5, tamoxifen could more "effectively kill several Leishmania species".
In the 2015 Coelho A et al. study, the researchers stated, "We have previously reported that tamoxifen, a selective estrogen receptor modulator, is active in vitro and in vivo against Leishmania species. We have demonstrated that tamoxifen's antileishmanial activity is not dependent on the interaction with estrogen receptors, but the precise antileishmanial mechanism of action is still uncertain ..... Tamoxifen must have multiple targets in the parasite in agreement with observations in tumor cells where, besides being able to bind and modulate estrogen receptor activity, tamoxifen has been shown to exert other effects such as interference in membranes, ceramide metabolism, calcium-calmodulin modulation and protein kinase C activity."
(See Miguel DC et al., Tamoxifen is Effective Against Leishmania and Induces a Rapid Alkalinization of Parasitophorous Vacuoles Harboring Leishmania Amazonensis Amastigotes, J Antimicrob Chemother, 2007 .... See Coelho A et al., Leishmania is Not Prone to Develop Resistance to Tamoxifen, International Journal for Parasitology, Drugs, and Drug Resistance, 2015.)
TAMOXIFEN SIDE EFFECTS
"It seems that memory impairment may be considered as an important side effect of TAM (tamoxifen)."
Tajik A et al., Activation of the Dorsal Hippocampal Nicotinic Acetylcholine Receptors Improves Tamoxifen-Induced Memory Retrieval Impairment in Adult Female Rats, Neuroscience, 2016.
TAMOXIFEN SIDE EFFECTS
UTERINE CANCER - EXCERPTS FROM THE AMERICAN COLLEGE OF OBSTETRICIANS AND GYNECOLOGISTS, 2014
Endometrial Cancer: "Most studies have found that the increased relative risk of developing endometrial cancer for women taking tamoxifen is two to three times higher than that of an age-matched population. The level of risk of endometrial cancer in women treated with tamoxifen is dose and time dependent."
Uterine Sarcomas: "Uterine sarcomas consisting of leiomyosarcoma, carcinosarcoma, high-grade endometrial stromal sarcoma, adenosarcoma, and sarcoma not otherwise specified, are rare and estimated to comprise 8% of all invasive uterine cancer cases. In a review of all National Surgical Adjuvant Breast and Bowel Project breast cancer treatment trials, the rate of sarcoma in women treated with tamoxifen was 17 per 100,000 patient years versus none in the placebo group . Similarly, in a separate trial of high-risk women without breast cancer taking tamoxifen as part of a breast cancer prevention trial with a median follow-up of 6.9 years, there were four sarcomas (17 per 100,000 patient years) in the tamoxifen group versus none in the placebo group. This is compared with the incidence of one to two per 100,000 patient years in the general population." (See American College of Obstetricians and Gynecologists, June 2014, http://www.acog.org/Resources-And-Publications/Committee-Opinions/Committee-on-Gynecologic-Practice/Tamoxifen-and-Uterine-Cancer.)
TAMOXIFEN SIDE EFFECTS
ULTRASOUND ENDOMETRIAL EVALUATION, HYSTEROSCOPY, AND HISTOPATHOLOGY IN BREAST CANCER WHILE ON TAMOXIFEN
"Most studies have found that the increased relative risk of developing endometrial cancer for women taking Tamoxifen is two to three times higher than that of an age-matched population."
In the 2015 Jindal A et al. study, the researchers investigated the health of the endometrium in breast cancer patients taking tamoxifen.
Out of fifty breast cancer patients chosen for ultrasonography, "35 patients (70%) had an endometrial thickness up to 5 mm. 15 patients (30%) had an endometrial thickness more than 5mm. Out of these, 11 patients, i.e. 22% of total, had an endometrial thickness of 5.1 to 10 mm and 2 patients, i.e. 4% of total had an endometrial thickness of more than 20 mm. Hysteroscopy was done on 11 patients. Out of these 8 patients had a normal hysteroscopic appearance whereas 3 patients had an abnormal hysteroscopic picture. Endometrial HPE (biopsy) of these 11 patients revealed 2 patients had secretory changes, 1 had polyp change, 1 had atrophic endometrium, 3 had simple endometrial hyperplasia, 1 had endometrial adenocarcinoma and 4 patients were reported to have scanty curetting."
Conclusion: "The duration of tamoxifen therapy turned out to have a relationship with the incidence of endometrial carcinoma." In addition, the length of time on tamoxifen had a bearing on symptoms. ..."But there was no correlation between the length of time on tamoxifen and endometrial thickness." (See Jindal A et al., Endometrial Evaluation by Ultrasonography, Hysteroscopy and Histopathology in Cases of Breast Carcinoma on Tamoxifen Therapy, J Midlife Health, 2015.)
TAMOXIFEN SIDE EFFECTS
CENTRAL NERVOUS SYSTEM
In the 2013 Chen HY et al. study, the researchers stated, "It is increasingly apparent that treatment with a variety of anticancer agents often is associated with adverse neurological consequences. Clinical studies indicate that exposure even to tamoxifen, a putatively benign antihormonal agent widely used in breast cancer treatment, causes cognitive dysfunction and changes in central nervous system metabolism, hippocampal volume, and brain structure."
Specifically, in the study, "We found that tamoxifen is toxic for a variety of central nervous system cell populations in vitro and also increased cell death in the corpus callosum and reduced cell division in the mouse subventricular zone, the hippocampal dentate gyrus, and the corpus callosum."
The researchers did find a way to "rescue" the central nervous system cells with a drug, MEK1/2, which"selectively rescued primary glial progenitors from tamoxifen toxicity in vitro while enhancing tamoxifen effects on MCF7 luminal human breast cancer cells. In vivo, MEK1/2 inhibition prevented tamoxifen-induced cell death in systemically treated mice."
Conclusion: "Our results demonstrate unexpected cytotoxicity of this putatively benign antihormonal agent (tamoxifen) and offer a potential strategy for rescuing central nervous systemcells from adverse effects of tamoxifen.
EDITORS' NOTE: We already know that chemo is associated with memory problems and that adding tamoxifen "may lead to more widespread memory deficits" (Bender CM et al., 2006). From the Chen HY et al. study, we have learned with specificity about the tamoxifen's toxicity in difference central nervous system cell populations. Since tamoxifen has exhibited this toxicity, should a breast cancer patient then need to chance another drug, MEK1/2, to rescue the cells from the toxicity?
(See Chen HY et al., MEK1/2 Inhibition Suppresses Tamoxifen Toxicity on CNS Glial Progenitor Cells, Journal of Neuroscience, 2013.)
Specifically, in the study, "We found that tamoxifen is toxic for a variety of central nervous system cell populations in vitro and also increased cell death in the corpus callosum and reduced cell division in the mouse subventricular zone, the hippocampal dentate gyrus, and the corpus callosum."
The researchers did find a way to "rescue" the central nervous system cells with a drug, MEK1/2, which"selectively rescued primary glial progenitors from tamoxifen toxicity in vitro while enhancing tamoxifen effects on MCF7 luminal human breast cancer cells. In vivo, MEK1/2 inhibition prevented tamoxifen-induced cell death in systemically treated mice."
Conclusion: "Our results demonstrate unexpected cytotoxicity of this putatively benign antihormonal agent (tamoxifen) and offer a potential strategy for rescuing central nervous systemcells from adverse effects of tamoxifen.
EDITORS' NOTE: We already know that chemo is associated with memory problems and that adding tamoxifen "may lead to more widespread memory deficits" (Bender CM et al., 2006). From the Chen HY et al. study, we have learned with specificity about the tamoxifen's toxicity in difference central nervous system cell populations. Since tamoxifen has exhibited this toxicity, should a breast cancer patient then need to chance another drug, MEK1/2, to rescue the cells from the toxicity?
(See Chen HY et al., MEK1/2 Inhibition Suppresses Tamoxifen Toxicity on CNS Glial Progenitor Cells, Journal of Neuroscience, 2013.)
TAMOXIFEN SIDE EFFECTS
GLUCOSE AND LIPID METABOLISM PARAMETERS, BROWNING OF SUBCUTANEOUS ADIPOSE TISSUE, TRANSIENT BODY CHANGES IN MICE
In the Hesselbarth N et al. 2015 study, the researchers, noting that estrogen receptor signaling plays an "important" role in adipose tissue metabolism, tested whether tamoxifen would change the metabolism of adipose tissue in mice.
Mice were given tamoxifen for five days, then tested for six weeks for tamoxifen's "effects on body composition, energy homeostasis, parameters of adipose tissue biology, glucose, and lipid metabolism".
Results: In the mice, the tamoxifen caused "significantly increased HbA1c, triglyceride and fatty
acid serum concentrations, browning of subcutaneous adipose tissue and increased UCP-1 (brown fat uncoupling protein) expression, increased adipose tissue proliferation marker Ki67 mRNA expression, changes in adipocyte size distribution, and transient body composition changes." (See Hesselbarth N et al.,Tamoxifen Affects Glucose and Lipid Metabolism Parameters, Causes Browning of Subcustaneous Adipose Tissue and Transient Body Composition Changes in C57BL/6NTac Mice, Biochem Biophys Res Commun, 2015.)
WHY TAMOXIFEN MAY NOT WORK
NEED TOTAL DARKNESS AT NIGHT - CIRCADIAN AND MELATONIN DISRUPTION BY LIGHT AT NIGHT
DRIVE RESISTANCE TO TAMOXIFEN THERAPY IN BREAST CANCER
In the Dauchy RT et al. 2014 study, the researchers explored tamoxifen resistance and night light's disruption of melatonin and circadian rhythms.
For their study, the researchers used a rat model of estrogen receptor-positive tumor xenografts to test light/dark cycles' impact on tumor growth, which may "confer an intrinsic resistance to tamoxifen therapy".
From the Tulane University Press Release, By Arthur Nead (2014) :
“ 'In the first phase of the study, we kept animals in a daily light/dark cycle of 12 hours of light followed by 12 hours of total darkness (melatonin is elevated during the dark phase) for several weeks,' says Steven Hill. 'In the second study, we exposed them to the same daily light/dark cycle; however, during the 12 hour dark phase, animals were exposed to extremely dim light at night (melatonin levels are suppressed), roughly equivalent to faint light coming under a door.'
Melatonin by itself delayed the formation of tumors and significantly slowed their growth, but tamoxifen caused a dramatic regression of tumors in animals with either high nighttime levels of melatonin during complete darkness or those receiving melatonin supplementation during dim light at night exposure.
These findings have potentially enormous implications for women being treated with tamoxifen and also regularly exposed to light at night due to sleep problems, working night shifts or exposed to light from computer and TV screens.
'High melatonin levels at night put breast cancer cells to 'sleep' by turning off key growth mechanisms. These cells are vulnerable to tamoxifen. But when the lights are on and melatonin is suppressed, breast cancer cells ‘wake up’ and ignore tamoxifen,' David Blask says."
Conclusion from study: "Strikingly, our results also showed that melatonin acted both as a tumor metabolic inhibitor and a circadian-regulated kinase inhibitor to reestablish the sensitivity of breast tumors to tamoxifen and tumor regression. Together, our findings show how dLEN (dim light)-mediated disturbances in nocturnal melatonin production can render tumors insensitive to tamoxifen."
(For the 2014 Tulane Press Release, see Arthur Nead,https://tulane.edu/news/releases/pr_072514.cfm .... For the study, see Dauchy RT et al., Circadian and Melatonin Disruption by Exposure to Light at Night Drives Intrinsic Resistance to Tamoxifen Therapy in Breast Cancer, Cancer Res, 2014.)
WHY TAMOXIFEN MAY NOT WORK
TAMOXIFEN DRUG RESISTANCE
"Tamoxifen generally blocks the binding process of the estrogen-receptor, but some estrogen receptor-positive cancers are resistant or become resistant to tamoxifen therapy, finding ways to elude its effects"
There is a gene, MACROD2, that has been identified as being a part of tamoxifen resistance.
"MACROD2 appears to code for a biological path to tamoxifen resistance by diverting the drug from its customary blocking process to a different way of latching onto breast cancer cell receptors, causing cancer cell growth rather than suppression ... Specifically, the team’s (Park BH et al.'s 2014) experiments found that when the gene is overexpressed in breast cancer cells—producing more of its protein product than normal—the cells become resistant to tamoxifen."
http://www.hopkinsmedicine.org/news/media/releases/johns_hopkins_scientists_link_gene_to_tamoxifen_resistant_breast_cancers.)
POSSIBLE TAMOXIFEN ALTERNATIVES - EXAMPLES OF NATURAL FOODS AND HERBS
TOTAL NIGHT DARKNESS, MELATONIN, AND POMEGRANATE
"...The PME (methanolic extract of pomegranate) has an anti-estrogenic effect on the mammary gland, without compromising the beneficial effects of estrogen in the cardiovascular and skeletal system and had no estrogenicity in the uterus. PME could possibly be considered as an ideal SERM (like tamoxifen) and further studies might demonstrate its suitability and possible application in estrogen dependent breast cancers with beneficial affects in other normal dependent tissues." (See Sreekumar S et al., Pomegranate as a Rich Source of Biologically Active Compounds, Biomed Res Int, 2014.)
EDITORS' NOTE: Just like with tamoxifen, why not try total darkness at night to sleep, take melatonin, and eat pomegranate to act like a SERM!
FROM OUR FOODS' SECTION: One of pomegranate's mechanisms of action: "Our earlier finding also demonstrated the role of proliferation in breast cancer by binding to estrogen receptor at the same time not affecting uterine weight unlike estradiol (a form of estrogen) or tamoxifen (a drug)." (See Ravindran and Sreeharshen, Punica granatum and its Therapeutic Implications on Breast Carcinogenesis: A Review, BioFactors, 2015.)
"We had found the PME (methanolic extract of pomegranate) has an anti-estrogenic effect on the mammary gland, without compromising the beneficial effects of estrogen in the cardiovascular and skeletal system and had no estrogenicity in the uterus. PME could possibly be considered as an ideal SERM and further studies might demonstrate its suitability and possible application in estrogen dependent breast cancers with beneficial affects in other normal dependent tissues....Furthermore, it would be valuable to investigate the long-term effects of PME in the in vivo models of estrogen deprivation to demonstrate its suitability in HRT (hormone replacement therapy). To achieve this goal, a better understanding is needed regarding the action of SERM, receptor, and coregulators that contribute to distinct patterns of gene expression." (See Sreekumar S et al., Pomegranate as a Rich Source of Biologically Active Compounds, Biomed Res Int, 2014.)
POSSIBLE TAMOXIFEN ALTERNATIVES - EXAMPLES OF NATURAL FOODS AND HERBS
TOTAL NIGHT DARKNESS, MELATONIN, AND EUGENOL (FROM CLOVES)
"..Compared to tamoxifen and its metabolites, eugenol (in cloves) can be considered as a small fragment that has a potency to be developed further in a direction guided by a computer-aided structure-bound design to have compounds that have similar or even better affinities to the estrogen receptor than tamoxifen and its metabolites." Anita Y et al, 2012.
EDITORS' NOTE: Just like with tamoxifen, why not try total darkness at night to sleep, take melatonin, and take cloves to act like a SERM!
In the Anita Y et al. 2012 study, the researchers investigated screening protocols for eugenol analogs to be used as potential estrogen antagonists.
"Eugenol is an essential oil mainly found in the buds and leaves of clove (Syzygium aromaticum (L.) which has been reported to have activity on inhibition of cell proliferation and apoptosis induction in human hormone receptor- positive breast cancer cells."
Further, "Tamoxifen itself is a prodrug that is metabolized in the liver results in some active metabolites (e.g., 4-hydroxytamoxifen and N-desmethyl-4-hydroxy-tamoxifen), with 30-100 fold activity in the binding to estrogen receptors compared to its original form .On the other hand, compared to tamoxifen and its metabolites, eugenol can be considered as a small fragment that has a potency to be developed further in a direction guided by a computer-aided structure-based design to have compounds that have similar or even better affinities to estrogen receptors than tamoxifen and its metabolites."
Conclusion: ". Based on the virtual screening results, dimer eugenol of 4-[4-hydroxy-3-(prop-2-en-1- yl)phenyl]-2-(prop-2-en-1-yl)phenol is recommended to be developed further in order to discover novel and potent ER antagonists." (See Structure-Based Design of Eugenol Analogs as Potential Estrogen Antagonists, Bioinformation, 2012.)
In the Anita Y et al. 2012 study, the researchers investigated screening protocols for eugenol analogs to be used as potential estrogen antagonists.
"Eugenol is an essential oil mainly found in the buds and leaves of clove (Syzygium aromaticum (L.) which has been reported to have activity on inhibition of cell proliferation and apoptosis induction in human hormone receptor- positive breast cancer cells."
Further, "Tamoxifen itself is a prodrug that is metabolized in the liver results in some active metabolites (e.g., 4-hydroxytamoxifen and N-desmethyl-4-hydroxy-tamoxifen), with 30-100 fold activity in the binding to estrogen receptors compared to its original form .On the other hand, compared to tamoxifen and its metabolites, eugenol can be considered as a small fragment that has a potency to be developed further in a direction guided by a computer-aided structure-based design to have compounds that have similar or even better affinities to estrogen receptors than tamoxifen and its metabolites."
Conclusion: ". Based on the virtual screening results, dimer eugenol of 4-[4-hydroxy-3-(prop-2-en-1- yl)phenyl]-2-(prop-2-en-1-yl)phenol is recommended to be developed further in order to discover novel and potent ER antagonists." (See Structure-Based Design of Eugenol Analogs as Potential Estrogen Antagonists, Bioinformation, 2012.)
POSSIBLE TAMOXIFEN ALTERNATIVES - EXAMPLES OF NATURAL FOODS AND HERBS
TOTAL NIGHT DARKNESS, MELATONIN, AND ARTEMISININ (FROM WORMWOOD) ALONG WITH FULVESTRANT (FASLODEX)
"Our studies suggest that artemisinin (from wormwood) treatment could potentially slow down ERα-induced proliferative signaling in low- and high-risk premalignant lesions and possibly the overall process of promotion of initiated cells to clinical breast cancer. As such, artemisinin could potentially be used in combinational therapies with well-established antiestrogens (like fulvestrant and tamoxifen).." ..Sundar S et al., 2008.
EDITORS' NOTE: Just like with tamoxifen, why not try total darkness at night to sleep, take melatonin, and take a combination of artemisinin and fulvestrant to act like a SERM!
In the Sundar S et al. 2012 study, the researchers observed that, "The majority of breast cancers expressing estrogen receptor α (ERa) are estrogen sensitive and are clinically managed with mixed non-steroidal antiestrogens such as tamoxifen, although detrimental side effects to long-term treatment with this antiestrogen include an increased endometrial cancer risk and eventual resistance. Pure steroidal antiestrogens, such as fulvestrant (Faslodex) are promising therapeutics for hormone-responsive breast cancer."
What about an anti-estrogenic natural plant compound?
"Natural plant compounds provide a potential source of such chemotherapeutic agents that act on various types of human breast cancers. One such promising natural compound is artemisinin, a sesquiterpene lactone that was isolated from a Chinese plant, Artemesia annua (wormwood)."
Artemisinin (natural plant compound) and fulvestrant (anti-estrogen)in combo?
Yes, when the researchers treated estrogen receptor-positive breast cancer cells with artemisinin alongside fulvestrant, there was "a cooperative reduction of the ERa protein levels and enhanced G1 cell cycle arrest compared with the effects of either compound alone. Our results show that artemisinin switches proliferative human breast cancer cells from expressing a high ERα:ERβ ratio to a condition in which ERβ predominates, which parallels the physiological state linked to antiproliferative events in normal mammary epithelium."
The researchers commented, "Our studies suggest that artemisinin treatment could potentially slow down ERα-induced proliferative signaling in low- and high-risk premalignant lesions and possibly the overall process of promotion of initiated cells to clinical breast cancer. As such, artemisinin could potentially be used in combinational therapies with well-established antiestrogens."
Conclusion:"Thus, we propose that artemisinin has the potential to be a strong candidate for adjuvant therapy with fulvestrant and could be extended to other breast cancer therapies such as tamoxifen. Patients could also benefit from lowering the systemic exposure of the patient to antiestrogens and minimizing undesirable side effects due to artemisinin–antiestrogen cooperativity." (See Sundar S et al., Artemisinin Selectively Decreases Functional Levels of Estrogen Receptor-Alpha and Ablates Estrogen-Induced Proliferation in Human Breast Cancer Cells, Oxford University Press, 2008.)
In the Sundar S et al. 2012 study, the researchers observed that, "The majority of breast cancers expressing estrogen receptor α (ERa) are estrogen sensitive and are clinically managed with mixed non-steroidal antiestrogens such as tamoxifen, although detrimental side effects to long-term treatment with this antiestrogen include an increased endometrial cancer risk and eventual resistance. Pure steroidal antiestrogens, such as fulvestrant (Faslodex) are promising therapeutics for hormone-responsive breast cancer."
What about an anti-estrogenic natural plant compound?
"Natural plant compounds provide a potential source of such chemotherapeutic agents that act on various types of human breast cancers. One such promising natural compound is artemisinin, a sesquiterpene lactone that was isolated from a Chinese plant, Artemesia annua (wormwood)."
Artemisinin (natural plant compound) and fulvestrant (anti-estrogen)in combo?
Yes, when the researchers treated estrogen receptor-positive breast cancer cells with artemisinin alongside fulvestrant, there was "a cooperative reduction of the ERa protein levels and enhanced G1 cell cycle arrest compared with the effects of either compound alone. Our results show that artemisinin switches proliferative human breast cancer cells from expressing a high ERα:ERβ ratio to a condition in which ERβ predominates, which parallels the physiological state linked to antiproliferative events in normal mammary epithelium."
The researchers commented, "Our studies suggest that artemisinin treatment could potentially slow down ERα-induced proliferative signaling in low- and high-risk premalignant lesions and possibly the overall process of promotion of initiated cells to clinical breast cancer. As such, artemisinin could potentially be used in combinational therapies with well-established antiestrogens."
Conclusion:"Thus, we propose that artemisinin has the potential to be a strong candidate for adjuvant therapy with fulvestrant and could be extended to other breast cancer therapies such as tamoxifen. Patients could also benefit from lowering the systemic exposure of the patient to antiestrogens and minimizing undesirable side effects due to artemisinin–antiestrogen cooperativity." (See Sundar S et al., Artemisinin Selectively Decreases Functional Levels of Estrogen Receptor-Alpha and Ablates Estrogen-Induced Proliferation in Human Breast Cancer Cells, Oxford University Press, 2008.)
AROMATASE INHIBITORS (AIs)
*POSTMENOPAUSAL WOMEN
ARE
GIVEN
THE ANTI-HORMONE
THERAPY,
AROMATASE INHIBITORS. *
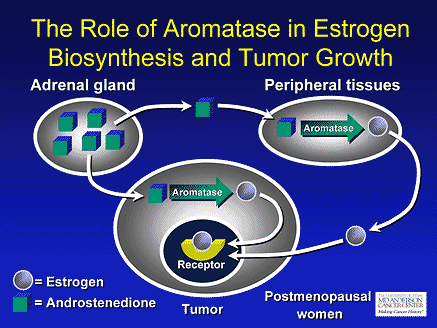
"An important concept is that postmenopausal women not only produce estrogens through peripheral tissues through the activity of the specific enzyme called aromatase, but also that aromatase is present in high concentrations in many breast cancers. The breast cancer cells or the stroma present in breast cancer cells has aromatase in high amounts, and you want to disrupt this or inhibit the production of estrogens in the target area where it counts most, and within the tumor deposit. This is something else that the aromatase inhibitors accomplish quite well." http://www.medscape.org/viewarticle/450047
AROMATASE INHIBITORS
"Aromatase inhibitors are drugs that lower estrogen levels by stopping an enzyme in fat tissue (called aromatase) from changing other hormones into estrogen. These drugs don’t stop the ovaries from making estrogen. Since most estrogen in the body is made by the ovaries, these drugs only lower estrogen levels in women whose ovaries aren’t making estrogen (such as those who have already gone through menopause). The drugs in this class include:
- Exemestane (Aromasin®)
- Letrozole (Femara®)
- Anastrozole (Arimidex®)
http://www.cancer.org/cancer/breastcancer/moreinformation/medicinestoreducebreastcancer/medicines-to-reduce-breast-cancer-risk-aromatase-inhibitors
AROMATASE INHBITORS ALONE OR SEQUENTIALLY COMBINED WITH TAMOXIFEN
VERSUS
TAMOXIFEN OR PLACEBO IN EARLY BREAST CANCER IN POSTMENOPAUSAL WOMEN
Highlights - Meta-Analysis, Ryder L et al., 2016 Study
•"Monotherapy with aromatase inhibitors improves disease free survival and overall survival compared with tamoxifen.
• AI sequentially combined with tamoxifen improves disease free survival compared with tamoxifen alone.
• The effect of sequenced therapy with aromatase inhibitors on disease free survival is not significant in studies with randomization before any endocrine therapy.
• Extended therapy with aromatase inhibitors improve disease-free survival.
• Cardiovascular events related to aromatase inhibitor treatment cannot be ruled out."
In the Ryder L et al. 2016 study, the researchers conducted, "A systematic review of seven randomized controlled studies comparing TAM (tamoxifen) and AIs (aromatase inhibitors), and one study comparing extended therapy with an AI with placebo after about 5 years of tamoxifen, aims to assess long-term clinical efficacy and adverse events."
Conclusion: "AIs are superior to TAM as adjuvant hormonal therapy for postmenopausal ER-positive breast cancer. TAM can be considered for individual patients due to the different toxicity profile compared with AI. Cardiovascular events related to AI treatment deserve further attention." (See Ryder L et al., Aromatase Inhibitors Alone or Sequentially Combined with Tamoxifen in Postmenopausal Early Breast Cancer Compared with Tamoxifen or Placebo - Meta-Analyses on Efficacy and Adverse Events Based on Randomized Clinical Trials, The Breast, 2016.)
Highlights - Meta-Analysis, Ryder L et al., 2016 Study
•"Monotherapy with aromatase inhibitors improves disease free survival and overall survival compared with tamoxifen.
• AI sequentially combined with tamoxifen improves disease free survival compared with tamoxifen alone.
• The effect of sequenced therapy with aromatase inhibitors on disease free survival is not significant in studies with randomization before any endocrine therapy.
• Extended therapy with aromatase inhibitors improve disease-free survival.
• Cardiovascular events related to aromatase inhibitor treatment cannot be ruled out."
In the Ryder L et al. 2016 study, the researchers conducted, "A systematic review of seven randomized controlled studies comparing TAM (tamoxifen) and AIs (aromatase inhibitors), and one study comparing extended therapy with an AI with placebo after about 5 years of tamoxifen, aims to assess long-term clinical efficacy and adverse events."
Conclusion: "AIs are superior to TAM as adjuvant hormonal therapy for postmenopausal ER-positive breast cancer. TAM can be considered for individual patients due to the different toxicity profile compared with AI. Cardiovascular events related to AI treatment deserve further attention." (See Ryder L et al., Aromatase Inhibitors Alone or Sequentially Combined with Tamoxifen in Postmenopausal Early Breast Cancer Compared with Tamoxifen or Placebo - Meta-Analyses on Efficacy and Adverse Events Based on Randomized Clinical Trials, The Breast, 2016.)
AIs VERSUS TAMOXIFEN SIDE EFFECTS
Aromatase inhibitors
Common side effects
Tamoxifen
Common side effects
http://ww5.komen.org/BreastCancer/SideEffectsofAromataseInhibitors.html
Common side effects
- "Hot flashes and night sweats
- Joint and muscle pain
- Loss of bone mineral density (may lead to osteoporosis or bone fractures)
- Loss of sex drive
- Vaginal dryness or itching
Tamoxifen
Common side effects
- Hot flashes and night sweats
- Loss of sex drive
- Vaginal discharge
- Vaginal dryness or itching"
http://ww5.komen.org/BreastCancer/SideEffectsofAromataseInhibitors.html