THERAPY - RADIATION THERAPY
ONE REASON WHY RADIATION THERAPY MAY FAIL - IN SOME CASES, IN BREAST CANCER RADIATION THERAPY - RADIATION INDUCES BREAST CANCER STEM CELLS!
" ....Radiation treatment -- despite killing half
of all tumor cells during every treatment --
transforms other cancer cells into treatment- resistant breast cancer stem cells." https://www.sciencedaily.com/releases/2012/02/120213185115.htm
CANCER STEM CELLS:
In 1889, Steven Paget theorized that cancer cells "within a tumor have the intrinsic capability to 'seed" a metastasis to a distant organ". Vlashi E et al., 2015
In general, stem cells are used for regeneration. Stem cells have been found to have lower ROS (reactive oxygen species) than other cells due to their high antioxidants to help protect them. "This may explain why cultured mouse breast cancer stem cells were less likely than other cancer cells to experience DNA damage after ionizing radiation." http://med.stanford.edu/news/all-news/2009/02/source-of-cancer-stem-cells-resistance-to-radiation-discovered-at-stanfo
CANCER STEM CELLS INITIATE TUMORS
Cancer stem cells are hypothesized to be at the apex of the cell's hierarchy. "In this model, the ability to initiate tumors and to give rise to the heterogeneous cell populations found in the original tumor is exclusively attributed to the cancer stem cell population with all of their differentiated progeny lacking these features." Vlashi E et al., 2015.
RADIATION THERAPY CAN INDUCE CANCER STEM CELLS:
In the Lagadec C et al. 2012 study, the researchers extracted the few breast cancer stem cells before using radiation upon the rest of the breast cancer cells. Then the irradiated cells were inserted into mice. Upshot? Induced breast cancer stem cells! Lagadec C et al., 2012.
These induced breast cancer stem cells have a more than 30-fold increased ability to form tumors!
And then add these induced breast cancer stem cells to the already existing breast cancer stem cells!
CONCLUSION: "Radiation may induce a breast cancer stem cell phenotype .... which contributes to increased cancer stem cell numbers..." Vlashi E et al., 2015.
of all tumor cells during every treatment --
transforms other cancer cells into treatment- resistant breast cancer stem cells." https://www.sciencedaily.com/releases/2012/02/120213185115.htm
CANCER STEM CELLS:
In 1889, Steven Paget theorized that cancer cells "within a tumor have the intrinsic capability to 'seed" a metastasis to a distant organ". Vlashi E et al., 2015
In general, stem cells are used for regeneration. Stem cells have been found to have lower ROS (reactive oxygen species) than other cells due to their high antioxidants to help protect them. "This may explain why cultured mouse breast cancer stem cells were less likely than other cancer cells to experience DNA damage after ionizing radiation." http://med.stanford.edu/news/all-news/2009/02/source-of-cancer-stem-cells-resistance-to-radiation-discovered-at-stanfo
CANCER STEM CELLS INITIATE TUMORS
Cancer stem cells are hypothesized to be at the apex of the cell's hierarchy. "In this model, the ability to initiate tumors and to give rise to the heterogeneous cell populations found in the original tumor is exclusively attributed to the cancer stem cell population with all of their differentiated progeny lacking these features." Vlashi E et al., 2015.
RADIATION THERAPY CAN INDUCE CANCER STEM CELLS:
In the Lagadec C et al. 2012 study, the researchers extracted the few breast cancer stem cells before using radiation upon the rest of the breast cancer cells. Then the irradiated cells were inserted into mice. Upshot? Induced breast cancer stem cells! Lagadec C et al., 2012.
These induced breast cancer stem cells have a more than 30-fold increased ability to form tumors!
And then add these induced breast cancer stem cells to the already existing breast cancer stem cells!
CONCLUSION: "Radiation may induce a breast cancer stem cell phenotype .... which contributes to increased cancer stem cell numbers..." Vlashi E et al., 2015.
"Radiation induces de novo generation of functional cancer stem cells."
Lagadec C et al;., 2012.
"(With radiation and chemo), there is a technicality of shrinking the size of the tumor, but enriching the population of tumorigenic cells (cancer stem cells)."
Sayer Ji, 2015.
"There is no such thing as a radiation exposure that will not do damage. There is a hundred per cent possibility that there will be damage to cells. The next question is: Which damage do you care about? The damage which is apt to cause most trouble in a whole system like a human being is the damage that hits the nucleus of the cell. Because inside the nucleus is the chromosome material that carries the template of what the cell does. If you change that, you change what the cell produces. If you change one cell, and it is still able to produce, it makes two cells with damaged chromosomes which can cause exponential growth of cells that are not going to do the right thing."
Sister Rosalie Bertell. PHD, Speaking in Oslo, Norway in 1990
Lagadec C et al;., 2012.
"(With radiation and chemo), there is a technicality of shrinking the size of the tumor, but enriching the population of tumorigenic cells (cancer stem cells)."
Sayer Ji, 2015.
"There is no such thing as a radiation exposure that will not do damage. There is a hundred per cent possibility that there will be damage to cells. The next question is: Which damage do you care about? The damage which is apt to cause most trouble in a whole system like a human being is the damage that hits the nucleus of the cell. Because inside the nucleus is the chromosome material that carries the template of what the cell does. If you change that, you change what the cell produces. If you change one cell, and it is still able to produce, it makes two cells with damaged chromosomes which can cause exponential growth of cells that are not going to do the right thing."
Sister Rosalie Bertell. PHD, Speaking in Oslo, Norway in 1990
"Radiation therapy may be offered either after breast-conserving surgery or a mastectomy to help destroy any remaining cancer cells and lower the chances that the cancer will return to the breast or nearby lymph nodes." Radiation is additionally given in cases where the cancer has spread "to other areas, for example, to the bones or the brain".(See http://www.cancer.org/cancer/breastcancer/detailedguide/breast-cancer-treating-radiation)
Hypofractionated Radiation: Following a lumpectomy, the whole breast may be irradiated by an external beam for around six weeks. "Hypofractionated and conventional whole breast irradiation consist of the same radiation volume; however, in hypofractionated treatment, the dosage of each individual session is higher, ultimately enabling patients to receive the same amount of radiation in a shorter period of type. Nonetheless, both treatments have the same impact on cancer cells."
(See Barros A,http://radiationtherapynews.com/2015/02/03/hypofractionated-whole-breast-irradiation-breast-cancer-patients/. For the study, see Bekelman J et al., Uptake and Costs of Hypofractionated vs Conventional Whole Breast Irradiation After Breast Conserving Surgery in the United States, 2008–2013, JAMA, 2014.)
Whole Breast Irradiation Versus Partial Breast Irradiation: "An alternative approach (to whole breast irradiation) is partial breast radiation therapy (PBRT), using brachytherapy. This approach targets only the breast tissues around the tumor bed and is administered over a short-course of five days." The advantages? "Compared to whole breast irradiation, PBRT results in more rapid recovery from cancer-related fatigue with improved restoration of quality of life after radiation therapy. Additionally, better quality of life predicts higher NKCA (natural killer cells) against tumor targets, emphasizing the importance of fostering quality of life for women undergoing adjuvant radiation therapy."
(See Albuquerque K et al., Impact of Partial Versus Whole Breast Radiation Therapy on Fatigue, Perceived Stress, Quality of Life and Natural Killer Cell Activity in Womenwith Breast Cancer, BMC, 2012.)
Hypofractionated Radiation: Following a lumpectomy, the whole breast may be irradiated by an external beam for around six weeks. "Hypofractionated and conventional whole breast irradiation consist of the same radiation volume; however, in hypofractionated treatment, the dosage of each individual session is higher, ultimately enabling patients to receive the same amount of radiation in a shorter period of type. Nonetheless, both treatments have the same impact on cancer cells."
(See Barros A,http://radiationtherapynews.com/2015/02/03/hypofractionated-whole-breast-irradiation-breast-cancer-patients/. For the study, see Bekelman J et al., Uptake and Costs of Hypofractionated vs Conventional Whole Breast Irradiation After Breast Conserving Surgery in the United States, 2008–2013, JAMA, 2014.)
Whole Breast Irradiation Versus Partial Breast Irradiation: "An alternative approach (to whole breast irradiation) is partial breast radiation therapy (PBRT), using brachytherapy. This approach targets only the breast tissues around the tumor bed and is administered over a short-course of five days." The advantages? "Compared to whole breast irradiation, PBRT results in more rapid recovery from cancer-related fatigue with improved restoration of quality of life after radiation therapy. Additionally, better quality of life predicts higher NKCA (natural killer cells) against tumor targets, emphasizing the importance of fostering quality of life for women undergoing adjuvant radiation therapy."
(See Albuquerque K et al., Impact of Partial Versus Whole Breast Radiation Therapy on Fatigue, Perceived Stress, Quality of Life and Natural Killer Cell Activity in Womenwith Breast Cancer, BMC, 2012.)
WANT TO DO RADIATION THERAPY?
BEFORE RADIATION THERAPY - DO RADIATION SENSITIVITY TESTS!
Testing the blood of UNTREATED breast cancer patients and healthy people, there was more baseline damage in the patients' immune systems. TESTING USING THREE ASSAYS:
*48% of breast cancer patients were radiosensitive.
*8% of healthy people. radiosensitive
RESEARCHERS SUGGEST: TEST FOR RADIOSENSITIVTY BEFORE RADIATION THERAPY!
LOU JL ET AL., 2008. SEE BELOW.
BEFORE RADIATION THERAPY - DO RADIATION SENSITIVITY TESTS!
Testing the blood of UNTREATED breast cancer patients and healthy people, there was more baseline damage in the patients' immune systems. TESTING USING THREE ASSAYS:
*48% of breast cancer patients were radiosensitive.
*8% of healthy people. radiosensitive
RESEARCHERS SUGGEST: TEST FOR RADIOSENSITIVTY BEFORE RADIATION THERAPY!
LOU JL ET AL., 2008. SEE BELOW.
THRESHOLD QUESTION: A CANDIDATE FOR RADIATION THERAPY? IMMUNE SYSTEMS OF UNTREATED BREAST CANCER PATIENTS ALREADY DAMAGED? TEST FOR RADIATION SENSITIVITY BEFORE RADIATION THERAPY?
Yes!
In the 2008 Lou JL et al. study, the researchers took blood samples from untreated breast cancer patients and healthy people and tested the samples before and after radiation.
Before Radiation: Blood samples from the untreated breast cancer patients revealed that they had significantly more immune problems (micronucleated cell frequency and micronucleus frequency that reflect damage and fragments) than healthy people.
After Radiation: In the irradiated blood samples, genetic damage to the lymphocytes (types of white blood immune cells) in the breast cancer patients "increased significantly as compared to the controls as detected in the three genetic assays." In the three genetic tests, the untreated breast cancer patients exhibited radiation sensitivity between 40% to 48%, while the healthy people were only 8% radiation sensitive.
While concluding that the difference in lympocyte radiation sensitivity between the untreated breast cancer patients and the healthy people was significant, the researchers also noted that there were "wide individual variations" in the untreated breast cancer patients' blood on the genetic tests. Therefore, the researchers suggested "that multiple assays should be used to assess the radiosensitivity of patients with breast cancer before therapy."
A wonderful study! (See Lou JL et al., Response of Lymphocytes to Radiation in Untreated Breast Cancer Patients as Detected with Three Different Genetic Assays, Biomed Environ scie, 2008.)
THRESHOLD QUESTION: A CANDIDATE FOR RADIATION THERAPY?
TESTING FOR BRCA1/BRCA2 MUTATIONS?
"BRCA1 and BRCA2 are tumor suppressor genes that help repair damaged DNA. When BRCA1 or BRCA2 become mutated, then this damage can not be corrected. These damaged cells are more likely to develop additional genetic alterations that can lead to cancer." (See http://www.cancer.gov/cancertopics/causes-prevention/genetics/brca-fact-sheet.)
Yes!
In a retrospective 2012 cohort study, Pijpe A et al. wrote that their study on BRCA1/2 adds to the literature: "In BRCA1/2 mutation carriers, exposure to diagnostic radiation before the age of 30 was associated with an increased risk of breast cancer, at dose levels considerably lower than those at which increases have been found in other cohorts exposed to radiation. While previous studies were based only on mammography or radiography, this large cohort study used estimates of an individual age specific cumulative breast dose from various diagnostic radiation procedures as a measure of total diagnostic radiation exposure. The results support the use of non-ionising radiation imaging techniques (such as MRI) for surveillance in young with BRCA1/2 mutations. In conclusion, in this large European study among BRCA1/2 mutation carriers, exposure to diagnostic radiation before age 30 was associated with an increased risk of breast cancer, at dose levels considerably lower than those at which increases have been found in other cohorts exposed to radiation. The results of this study support the recommendation to use non-ionising radiation imaging techniques (such as MRI) as the main tool for surveillance in young BRCA1/2 mutation carriers."
(See Pijpe A et al., Exposure to Diagnostic Radiation and Risk of Breast Cancer Among Carriers of BRCA1/2 Mutations: Retrospective Cohort Study -GENE-RAD-RISK, BMJ, 2012.)
In a retrospective 2012 cohort study, Pijpe A et al. wrote that their study on BRCA1/2 adds to the literature: "In BRCA1/2 mutation carriers, exposure to diagnostic radiation before the age of 30 was associated with an increased risk of breast cancer, at dose levels considerably lower than those at which increases have been found in other cohorts exposed to radiation. While previous studies were based only on mammography or radiography, this large cohort study used estimates of an individual age specific cumulative breast dose from various diagnostic radiation procedures as a measure of total diagnostic radiation exposure. The results support the use of non-ionising radiation imaging techniques (such as MRI) for surveillance in young with BRCA1/2 mutations. In conclusion, in this large European study among BRCA1/2 mutation carriers, exposure to diagnostic radiation before age 30 was associated with an increased risk of breast cancer, at dose levels considerably lower than those at which increases have been found in other cohorts exposed to radiation. The results of this study support the recommendation to use non-ionising radiation imaging techniques (such as MRI) as the main tool for surveillance in young BRCA1/2 mutation carriers."
(See Pijpe A et al., Exposure to Diagnostic Radiation and Risk of Breast Cancer Among Carriers of BRCA1/2 Mutations: Retrospective Cohort Study -GENE-RAD-RISK, BMJ, 2012.)
THRESHOLD QUESTION: A CANDIDATE FOR RADIATION THERAPY? PREGNANT? TAKING PROGESTERONE? CAN PROGESTERONE INCREASE RADIATION DAMAGE?
Yes!
Wonder why a pregnant woman can't have X-rays? Likely due to increased progesterone.
In the Vares G et al. 2004 study, the researchers commented that while "Sex steroid hormones play an essential role in the control of homeostasis in the mammary gland," and knowing that "dysregulation of the apoptotic (cell death) plays an important role in the pathogenesis of breast cancer, we investigated the regulation of apoptosis by progesterone in various breast cancer cell lines."
Results? "...Progesterone treatment protects against radiation-induced apoptosis."
Import? "....Progesterone modifies the cellular radiation sensitivity by inhibiting the apoptotic process, allowing the survival and proliferation of DNA-damaged cells, and thus potentially facilitates the emergence of genetic mutations that may play a part in malignant transformation."
(See Vares G et al., Progesterone Prevents Radiation-Induced Apoptosis in Breast Cancer Cells, Oncogene, 2004.)
Wonder why a pregnant woman can't have X-rays? Likely due to increased progesterone.
In the Vares G et al. 2004 study, the researchers commented that while "Sex steroid hormones play an essential role in the control of homeostasis in the mammary gland," and knowing that "dysregulation of the apoptotic (cell death) plays an important role in the pathogenesis of breast cancer, we investigated the regulation of apoptosis by progesterone in various breast cancer cell lines."
Results? "...Progesterone treatment protects against radiation-induced apoptosis."
Import? "....Progesterone modifies the cellular radiation sensitivity by inhibiting the apoptotic process, allowing the survival and proliferation of DNA-damaged cells, and thus potentially facilitates the emergence of genetic mutations that may play a part in malignant transformation."
(See Vares G et al., Progesterone Prevents Radiation-Induced Apoptosis in Breast Cancer Cells, Oncogene, 2004.)
RADIATION THERAPY AFTER LUMPECTOMY?
A DISCUSSION: EXCERPTS - DO WE NEED TO IRRADIATE ALL SMALL INVASIVE BREAST CANCERS AND DCIS?
RADIATION THERAPY POST-LUMPECTOMY?
There is a " highly significant reduction in cancer recurrence (typically 70% to 75%) in the radiation-treated breast", without a "significant improvement in breast cancer survival".
Patients who are, "Post-lumpectomy with an anticipated ≤ 10% absolute reduction in locoregional risk by 5 years and overall recurrence (locoregional and distant) by 10 years can omit breast radiotherapy without risk of excess breast cancer mortality. Furthermore, older patients with node-negative, less than 2 cm, ER positive, low grade breast cancer who have taken tamoxifen characterize this lower risk group." Julia White, MD, 2013
There is a " highly significant reduction in cancer recurrence (typically 70% to 75%) in the radiation-treated breast", without a "significant improvement in breast cancer survival".
Patients who are, "Post-lumpectomy with an anticipated ≤ 10% absolute reduction in locoregional risk by 5 years and overall recurrence (locoregional and distant) by 10 years can omit breast radiotherapy without risk of excess breast cancer mortality. Furthermore, older patients with node-negative, less than 2 cm, ER positive, low grade breast cancer who have taken tamoxifen characterize this lower risk group." Julia White, MD, 2013
Julia White, MD ASCO University Book, 2013
"For Radiation After Lumpectomy: " ....Each of these trials demonstrated a highly significant reduction in cancer recurrence (typically 70% to 75%) in the treated breast with addition of breast radiotherapy. Despite fairly large reductions in local recurrence, none of these trials individually documented a significant improvement in breast cancer survival from radiotherapy after lumpectomy. However, on meta-analyses of these trials, reduction in local regional recurrence (LRR) after lumpectomy by radiotherapy resulted in a reduction in breast cancer mortality. This was demonstrated in the Early Breast Cancer Trialists Group (EBCTCG) meta-analysis that studied the relationship of local regional recurrence at 5 years and breast cancer mortality at 15 years in 7,311 patient level data from 10 randomized clinical trials. The 5-year local recurrence was 7% among those allocated radiotherapy and 26% among those observed, corresponding to an absolute reduction of 19%. The 15-year risk of death as a result of breast cancer was 30.5% among those allocated post-lumpectomy radiotherapy and 35.9% among those observed (corresponding to an absolute breast cancer mortality reduction of 5.4%, SE 1·7). The magnitude in reduction of breast cancer mortality is much smaller than that for local recurrence but still a highly significant breast cancer death rate ratio of 0.83, SE 0.05, 95% CI 0.75–0.91, 2, p = 0.0002. Subgroup analyses demonstrated that the absolute benefit produced by radiotherapy was determined principally by the magnitude of the local recurrence risk in unirradiated women. In particular, an analyses that divided absolute local recurrence risk reduction after lumpectomy or mastectomy by 5 years into three categories of less than 10%, 10% to 20%, or greater than 20% demonstrated that for those with less than 10% absolute reduction in local recurrence by 5 years no improvement in breast cancer mortality by 15 years is seen. A more recent meta-analysis from the EBCTCG of 10,801 women in 17 trials further explored the effect of post-lumpectomy breast radiotherapy on 10-year overall breast cancer recurrence rate (locoregional or distant) and 15-year breast cancer mortality effects.The 10-year risk of any (locoregional or distant) first recurrence was 19.3% in women allocated to radiotherapy and 35.0% in women allocated to breast-conserving surgery only, corresponding to an absolute risk reduction of 15.7% (95% CI 13.7–17.7, 2p<0.00001). Radiotherapy also reduced breast cancer death at 15 years by a moderate amount: 25.2% without and 21.4% with radiotherapy, respectively, for 15-year absolute risk reduction of 3.8% (95% CI 1.6–6.0, 2p = 0.00005).
Analysis of 7,287 pathologically node-negative patients by patient and tumor characteristics revealed that the absolute recurrence reduction varied strongly by patient age, tumor grade, tumor size, and ER status. Older women and those with low-grade tumors had a smaller risk of recurrence without radiotherapy than younger women and those with high-grade tumors. Each woman with pN0 disease was assigned an individually predicted absolute reduction in 10-year recurrence risk from radiotherapy that was categorized into three risk groups: lower (< 10%), intermediate (10% to 19%), and large (> 20%). The 10-year recurrence risks without radiotherapy was 50.3%, 24.8%, 18.9%, compared with 26%, 12.4%, and 12% (absolute reductions of 24.3%, 12.4%, and 6.9%) with radiotherapy for patients in the large, intermediate, and lower risk groups, respectively. The corresponding absolute reductions in 15-year risk of breast cancer death in the three groups were 7.8% (95% CI 3.1–12.5), 1.1% (−2.0 to 4.2), and 0.1% (−7.5 to 7.7), respectively. These analyses suggest that patients with breast cancer post-lumpectomy with an anticipated ≤ 10% absolute reduction in locoregional risk by 5 years and overall recurrence (locoregional and distant) by 10 years can omit breast radiotherapy without risk of excess breast cancer mortality. Furthermore, older patients with node-negative, less than 2 cm, ER positive, low grade breast cancer who have taken tamoxifen characterize this lower risk group " (SEE THE NEXT SECTION FOR A 2015 THE LANCET ONCOLOGY STUDY. For this Article Summary, See Do We Need to Irradiate All Small Invasive Breast Cancers and DCIS?, ASCO University Book, 2013.)
"For Radiation After Lumpectomy: " ....Each of these trials demonstrated a highly significant reduction in cancer recurrence (typically 70% to 75%) in the treated breast with addition of breast radiotherapy. Despite fairly large reductions in local recurrence, none of these trials individually documented a significant improvement in breast cancer survival from radiotherapy after lumpectomy. However, on meta-analyses of these trials, reduction in local regional recurrence (LRR) after lumpectomy by radiotherapy resulted in a reduction in breast cancer mortality. This was demonstrated in the Early Breast Cancer Trialists Group (EBCTCG) meta-analysis that studied the relationship of local regional recurrence at 5 years and breast cancer mortality at 15 years in 7,311 patient level data from 10 randomized clinical trials. The 5-year local recurrence was 7% among those allocated radiotherapy and 26% among those observed, corresponding to an absolute reduction of 19%. The 15-year risk of death as a result of breast cancer was 30.5% among those allocated post-lumpectomy radiotherapy and 35.9% among those observed (corresponding to an absolute breast cancer mortality reduction of 5.4%, SE 1·7). The magnitude in reduction of breast cancer mortality is much smaller than that for local recurrence but still a highly significant breast cancer death rate ratio of 0.83, SE 0.05, 95% CI 0.75–0.91, 2, p = 0.0002. Subgroup analyses demonstrated that the absolute benefit produced by radiotherapy was determined principally by the magnitude of the local recurrence risk in unirradiated women. In particular, an analyses that divided absolute local recurrence risk reduction after lumpectomy or mastectomy by 5 years into three categories of less than 10%, 10% to 20%, or greater than 20% demonstrated that for those with less than 10% absolute reduction in local recurrence by 5 years no improvement in breast cancer mortality by 15 years is seen. A more recent meta-analysis from the EBCTCG of 10,801 women in 17 trials further explored the effect of post-lumpectomy breast radiotherapy on 10-year overall breast cancer recurrence rate (locoregional or distant) and 15-year breast cancer mortality effects.The 10-year risk of any (locoregional or distant) first recurrence was 19.3% in women allocated to radiotherapy and 35.0% in women allocated to breast-conserving surgery only, corresponding to an absolute risk reduction of 15.7% (95% CI 13.7–17.7, 2p<0.00001). Radiotherapy also reduced breast cancer death at 15 years by a moderate amount: 25.2% without and 21.4% with radiotherapy, respectively, for 15-year absolute risk reduction of 3.8% (95% CI 1.6–6.0, 2p = 0.00005).
Analysis of 7,287 pathologically node-negative patients by patient and tumor characteristics revealed that the absolute recurrence reduction varied strongly by patient age, tumor grade, tumor size, and ER status. Older women and those with low-grade tumors had a smaller risk of recurrence without radiotherapy than younger women and those with high-grade tumors. Each woman with pN0 disease was assigned an individually predicted absolute reduction in 10-year recurrence risk from radiotherapy that was categorized into three risk groups: lower (< 10%), intermediate (10% to 19%), and large (> 20%). The 10-year recurrence risks without radiotherapy was 50.3%, 24.8%, 18.9%, compared with 26%, 12.4%, and 12% (absolute reductions of 24.3%, 12.4%, and 6.9%) with radiotherapy for patients in the large, intermediate, and lower risk groups, respectively. The corresponding absolute reductions in 15-year risk of breast cancer death in the three groups were 7.8% (95% CI 3.1–12.5), 1.1% (−2.0 to 4.2), and 0.1% (−7.5 to 7.7), respectively. These analyses suggest that patients with breast cancer post-lumpectomy with an anticipated ≤ 10% absolute reduction in locoregional risk by 5 years and overall recurrence (locoregional and distant) by 10 years can omit breast radiotherapy without risk of excess breast cancer mortality. Furthermore, older patients with node-negative, less than 2 cm, ER positive, low grade breast cancer who have taken tamoxifen characterize this lower risk group " (SEE THE NEXT SECTION FOR A 2015 THE LANCET ONCOLOGY STUDY. For this Article Summary, See Do We Need to Irradiate All Small Invasive Breast Cancers and DCIS?, ASCO University Book, 2013.)
EARLY BREAST CANCER? LUMPECTOMY? OVER 65?
SKIP RADIATION THERAPY?
RADIATION THERAPY
POST-LUMPECTOMY? OVER 65? RECEIVING HORMONE TREATMENT?
" in low risk older women receiving hormone treatment (the benefit of radiation therapy) is very modest. This makes omission of radiotherapy an option for selected older patients.."
Kunkler I et al., 2015
POST-LUMPECTOMY? OVER 65? RECEIVING HORMONE TREATMENT?
" in low risk older women receiving hormone treatment (the benefit of radiation therapy) is very modest. This makes omission of radiotherapy an option for selected older patients.."
Kunkler I et al., 2015
YES! AN OPTION!
"Some older women with breast cancer could safely avoid radiotherapy, without harming their chances of survival, a study has shown. Older women with early breast cancer who are given breast-conserving surgery and hormone therapy gain very modest benefit from radiotherapy, researchers conclude."
The lead researcher of the 2015 study, "Professor Ian Kunkler, of the University of Edinburgh's Cancer Research Center, commented: 'While radiotherapy will remain the standard of care for most women after breast-conserving surgery, the absolute reduction in risk of recurrence from radiotherapy in low risk older women receiving hormone treatment is very modest. This makes omission of radiotherapy an option for selected older patients.' "
(See http://www.sciencedaily.com/releases/2015/01/150128093544.htm. For the study, see Kunkler I et al., Breast-Conserving Surgery With or Without Irradiation in Women Aged 65 Years or Older With Early Breast Cancer (PRIMEII): A Randomized Controlled Trail, The Lancet Oncology, 2015.)
"Some older women with breast cancer could safely avoid radiotherapy, without harming their chances of survival, a study has shown. Older women with early breast cancer who are given breast-conserving surgery and hormone therapy gain very modest benefit from radiotherapy, researchers conclude."
The lead researcher of the 2015 study, "Professor Ian Kunkler, of the University of Edinburgh's Cancer Research Center, commented: 'While radiotherapy will remain the standard of care for most women after breast-conserving surgery, the absolute reduction in risk of recurrence from radiotherapy in low risk older women receiving hormone treatment is very modest. This makes omission of radiotherapy an option for selected older patients.' "
(See http://www.sciencedaily.com/releases/2015/01/150128093544.htm. For the study, see Kunkler I et al., Breast-Conserving Surgery With or Without Irradiation in Women Aged 65 Years or Older With Early Breast Cancer (PRIMEII): A Randomized Controlled Trail, The Lancet Oncology, 2015.)
POST-MASTECTOMY RADIATION THERAPY?
MAYBE SKIP RADIATION IF SPREAD TO JUST ONE LYMPH NODE?
RADIATION THERAPY? POST-MASTECTOMY?
"For the overwhelming majority of early-stage breast cancer patients treated with modern surgery and systemic therapies, locoregional recurrence rates may be too low to justify routine use of post-mastectomy radiation."
MD Anderon Press Release, 2010.
"For the overwhelming majority of early-stage breast cancer patients treated with modern surgery and systemic therapies, locoregional recurrence rates may be too low to justify routine use of post-mastectomy radiation."
MD Anderon Press Release, 2010.
Henry Kuerer, MD, PHD, Professor and Training Program Director in MD Anderson's Department of Surgical Oncology, said,
"For the overwhelming majority of early-stage breast cancer patients treated with modern surgery and systemic therapies, locoregional recurrence rates may be too low to justify routine use of post-mastectomy radiation. .... This research will provoke much discussion among those caring for women with early-stage breast disease. Replicating these findings should be a priority to ensure that patients only receive therapy that is medically necessary."
(See http://www.mdanderson.org/newsroom/news-releases/2010/most-early-stage-breast-cancer-patients-may-not-need-radiation-after-mastectomy.html/)
"For the overwhelming majority of early-stage breast cancer patients treated with modern surgery and systemic therapies, locoregional recurrence rates may be too low to justify routine use of post-mastectomy radiation. .... This research will provoke much discussion among those caring for women with early-stage breast disease. Replicating these findings should be a priority to ensure that patients only receive therapy that is medically necessary."
(See http://www.mdanderson.org/newsroom/news-releases/2010/most-early-stage-breast-cancer-patients-may-not-need-radiation-after-mastectomy.html/)
* EXPLORING MORE REASONS WHY RADIATION THERAPY MAY FAIL *
"Just taking breast cancer cells and injecting them into the previously irradiated, non-cancerous, normal thigh tissue of mice 'enhances the invasive capacity' of the breast cancer cells."
Lemay R , Irradiation of Normal Mouse Tissue Increases the Invasiveness of Mammary Cancer Cells, Int J Radiol Biol, 2011.
"Just taking breast cancer cells and injecting them into the previously irradiated, non-cancerous, normal thigh tissue of mice 'enhances the invasive capacity' of the breast cancer cells."
Lemay R , Irradiation of Normal Mouse Tissue Increases the Invasiveness of Mammary Cancer Cells, Int J Radiol Biol, 2011.
STARTING WITH MYC - DOES MYC HAVE A ROLE IN RADIATION-INDUCED BREAST CANCER?
Dr. James Watson, of Watson and Crick fame, urged us to study MYC's role in cancer. "Lots of MYC may turn out to be an essential feature of much of the truly incurable cancer. It simultaneously turns up the synthesis of the more than 1000 different proteins required to move all cells through the cell cycle. Although precisely how this almost 400-amino acid long polypeptide works at the molecular level remains to be worked out, it seems to play a unique role that cannot be handled by any other class of transcription factors."
Dr. Jim Watson , Oxidants, Antioxidants, and the Current Incurability of Metastatic Cancers, 2013.)
In Wade MA et al's 2014 study, the researchers explored MYC's role in radiation-induced breast cancer. "This study demonstrates that ionizing radiation directly induce genetic alterations affecting MYC, including translocation and high-level focal amplification and identified MYC as a radiosensitive locus. Importantly, MYC protein expression was significantly elevated in radiation-exposed breast cells, emphasizing the importance of this critical transcription faction in radiogenic breast cancer. Our findings provide insight into the etiology of radiogenic breast cancer and may have relevance to other cancers with a radiation exposure etiology."
(See Wade MA et al., c-MYC is a Radiosensitive Locus in Human Breast Cells, Oncogene, 2014.)
Dr. Jim Watson , Oxidants, Antioxidants, and the Current Incurability of Metastatic Cancers, 2013.)
In Wade MA et al's 2014 study, the researchers explored MYC's role in radiation-induced breast cancer. "This study demonstrates that ionizing radiation directly induce genetic alterations affecting MYC, including translocation and high-level focal amplification and identified MYC as a radiosensitive locus. Importantly, MYC protein expression was significantly elevated in radiation-exposed breast cells, emphasizing the importance of this critical transcription faction in radiogenic breast cancer. Our findings provide insight into the etiology of radiogenic breast cancer and may have relevance to other cancers with a radiation exposure etiology."
(See Wade MA et al., c-MYC is a Radiosensitive Locus in Human Breast Cells, Oncogene, 2014.)
RADIATION-INDUCED BREAST CANCER STEM CELLS - RADIATION INDUCES STEMNESS!!!
What are cancer stem cells? "Cancer stem cells are thought to represent a small sub-population of cells present in most tumors that, similar to normal tissue stem cells, possess the ability to self-renew, to divide asymmetrically and symmetrically, and to undergo multi-lineage differentiation." Cancer stem cells can "sustain tumors." Plus, these cancer stem cells are "believed to play a key role in cancer metastasis, cancer recurrence, and cancer drug resistance."
In the Ghisolfi L et al. 2012 study, the researchers "demonstrated that cancer cells can be induced by gamma radiation to acquire a stemness state characterized by increased stemness gene expression and a cancer stem cell-like phenotype."
How?
....By creating in non-stem cancer cells spherogenesis, and upregulating two pluripotency genes, SOX2 and Oct3/4.
Thus, "These data demonstrate that ionizing radiation can activate stemness pathways in heterogeneous cancer cells, resulting in the enrichment of a cancer stem cell subpopulation with higher resistance to radiation."
(See Ghisolfi L et al., Ionizing Radiation Induces Stemness in Cancer Cells, PLOS ONE, 2012.)
How?
....By creating in non-stem cancer cells spherogenesis, and upregulating two pluripotency genes, SOX2 and Oct3/4.
Thus, "These data demonstrate that ionizing radiation can activate stemness pathways in heterogeneous cancer cells, resulting in the enrichment of a cancer stem cell subpopulation with higher resistance to radiation."
(See Ghisolfi L et al., Ionizing Radiation Induces Stemness in Cancer Cells, PLOS ONE, 2012.)
PROGESTERONE GENERATES BREAST CANCER STEM CELLS IN IRRADIATED HUMAN MAMMARY CELL LINES
In the 2014 AACR Abstract, Vares G et al. investigated "whether the modulation of radiation-induced breast cancer risk by steroid hormones could involve cancer stem cells." To this end, the researchers evaluated the proportion of cancer stem cells in irradiated breast cancer cells lines after the introduction of estrogen and production, and found that the progesterone treatment and irradiation triggered cancer and stemness-associated micro RNA regulations,,,,, which resulted in increased proportion of radiation-resistant tumor-initiating cancer stem cells.
(See Vares G et al., Abstract 3856: Progesterone Generates Breast Cancer Stem Cells Through Nuclear Receptor Independent Mechanism in Irradiated Human Mammary Cell Lines, AACR; Cancer Res, 2014.)
DOES RADIATION THERAPY TRANSFORM LESS AGGRESSIVE BREAST CANCER CELLS INTO TREATMENT-RESISTANT CANCER STEM CELLS? MORE THAN A 30-FOLD INCREASE IN ABILITY TO FORM TUMORS? (CITED AT THE BEGINNING OF THE SECTION IN THE GREEN BOX.)
CANCER STEM CELLS NEED TO BE ERADICATED TO STAVE OFF RECURRENCE
"Breast cancer stem cells are thought to be the sole source of tumor recurrence and are known to be resistant to radiation therapy and don’t respond well to chemotherapy. Researchers report for the first time that radiation treatment -- despite killing half of all tumor cells during every treatment -- transforms other cancer cells into treatment-resistant breast cancer stem cells."
http://www.sciencedaily.com/releases/2012/02/120213185115.htm
"Breast cancer stem cells are thought to be the sole source of tumor recurrence and are known to be resistant to radiation therapy and don’t respond well to chemotherapy. Researchers report for the first time that radiation treatment -- despite killing half of all tumor cells during every treatment -- transforms other cancer cells into treatment-resistant breast cancer stem cells."
http://www.sciencedaily.com/releases/2012/02/120213185115.htm
"Their findings (Lagadec C et al., 2012) show that if tumors are challenged by certain stressors that threaten them (such as radiation), they generate induced breast cancer stem cells that may, along with surviving cancer stem cells, produce more tumors."
Carrie Printz, Radiation Treatment Generates Therapy-Resistant Cancer Stem Cells from Less Aggressive Breast Cancer Cells, Cancer, 2012.
Yes!
Radiation pushes less aggressive cells into a stem cell state, resulting in more than a 30- fold increased ability to form tumors!
From Science Daily: "Breast cancer stem cells are thought to be the sole source of tumor recurrence and are known to be resistant to radiation therapy and don’t respond well to chemotherapy. Researchers report for the first time that radiation treatment -- despite killing half of all tumor cells during every treatment -- transforms other cancer cells into treatment-resistant breast cancer stem cells."
An explanation in Science Daily: "We found that these induced breast cancer stem cells (iBCSC) were generated by radiation-induced activation of the same cellular pathways used to reprogram normal cells into induced pluripotent stem cells (iPS) in regenerative medicine," said Dr. Frank Pajonk, a radiation oncologist, who also is a scientist with the Eli and Edythe Broad Center of Regenerative Medicine at UCLA. "It was remarkable that these breast cancers used the same reprogramming pathways to fight back against the radiation treatment."
And, further, "The team also found that the iBCSC had a more than 30-fold increased ability to form tumors compared to the non-irradiated breast cancer cells from which they originated."... This study "suggests that undisturbed, growing tumors maintain a small number of cancer stem cells. However, if challenged by various stressors that threaten their numbers, including ionizing radiation, the breast cancer cells generate iBCSC that may, together with the surviving cancer stem cells, repopulate the tumor."
(See http://www.sciencedaily.com/releases/2012/02/120213185115.htm. For the study, see Lagadec C et al., Radiation-Induced Reprogramming of Breast Cancer Cells, Stem Cells, 2012.)
Radiation pushes less aggressive cells into a stem cell state, resulting in more than a 30- fold increased ability to form tumors!
From Science Daily: "Breast cancer stem cells are thought to be the sole source of tumor recurrence and are known to be resistant to radiation therapy and don’t respond well to chemotherapy. Researchers report for the first time that radiation treatment -- despite killing half of all tumor cells during every treatment -- transforms other cancer cells into treatment-resistant breast cancer stem cells."
An explanation in Science Daily: "We found that these induced breast cancer stem cells (iBCSC) were generated by radiation-induced activation of the same cellular pathways used to reprogram normal cells into induced pluripotent stem cells (iPS) in regenerative medicine," said Dr. Frank Pajonk, a radiation oncologist, who also is a scientist with the Eli and Edythe Broad Center of Regenerative Medicine at UCLA. "It was remarkable that these breast cancers used the same reprogramming pathways to fight back against the radiation treatment."
And, further, "The team also found that the iBCSC had a more than 30-fold increased ability to form tumors compared to the non-irradiated breast cancer cells from which they originated."... This study "suggests that undisturbed, growing tumors maintain a small number of cancer stem cells. However, if challenged by various stressors that threaten their numbers, including ionizing radiation, the breast cancer cells generate iBCSC that may, together with the surviving cancer stem cells, repopulate the tumor."
(See http://www.sciencedaily.com/releases/2012/02/120213185115.htm. For the study, see Lagadec C et al., Radiation-Induced Reprogramming of Breast Cancer Cells, Stem Cells, 2012.)
CAN NON-STEM CANCER CELLS RE-ACQUIRE CANCER STEM CELL TRAITS? FROM RADIATION THERAPY?
There is "evidence supporting the idea that non-stem cancer cells exhibit a remarkable degree of plasticity that allows them to re-acquire cancer stem cell traits, especially in the context of radiation therapy".
Vlashi E et al., 2015.
Vlashi E et al., 2015.
YES!
In the Vlashi E et al. 2015 study, the researchers note that for any cancer therapy to be successful, all of the small population of cancer stem cells, propelling the cancer, need to be killed off. On the other hand, the progeny of the cancer stem cells are differentiated cells that do not drive tumor growth, and their survival is "less critical". But, what happens when there is radiation therapy? There is "evidence supporting the idea that non-stem cancer cells exhibit a remarkable degree of plasticity that allows them to re-acquire cancer stem cell traits, especially in the context of radiation therapy."
(See Vlashi E et al.,Cancer Stem Cells, Cancer Cell Plasticity and Radiation Therapy, Semin Cancer Biol, 2015.)
In the Vlashi E et al. 2015 study, the researchers note that for any cancer therapy to be successful, all of the small population of cancer stem cells, propelling the cancer, need to be killed off. On the other hand, the progeny of the cancer stem cells are differentiated cells that do not drive tumor growth, and their survival is "less critical". But, what happens when there is radiation therapy? There is "evidence supporting the idea that non-stem cancer cells exhibit a remarkable degree of plasticity that allows them to re-acquire cancer stem cell traits, especially in the context of radiation therapy."
(See Vlashi E et al.,Cancer Stem Cells, Cancer Cell Plasticity and Radiation Therapy, Semin Cancer Biol, 2015.)
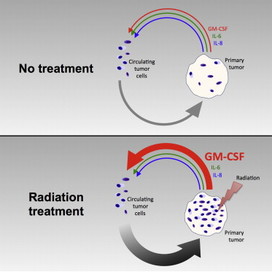
CIRCULATING CANCER TUMOR CELLS (CTCs) AND CANCER RADIATION - CAN RADIATION ATTRACT SHEDDING CANCER CELLS BACK TO THE TUMOR?
"Localized radiation therapy for breast cancer kills cancer cells at the tumor site. But, in a cruel irony, Cleveland Graves, PHD, and research assistant, Marta Vilalta, PHD, have found that the dying cells in the breast may send out a signal that recruits other cancer cells (circulating tumor cells) back to the side of the initial tumor."
Krista Conger, Radiation Therapy May Attract Circulating Tumor Cells, Stanford Scope, 2014.
Krista Conger, Radiation Therapy May Attract Circulating Tumor Cells, Stanford Scope, 2014.
SUMMARY OF VILALTA M ET AL. 2014 STUDY
- "•Radiation of tumor cells in vitro and in vivo attracts migrating tumor cells
- •Expression of GM-CSF (granulocyte-macrophage colony stimulating factor) is stimulated by radiation
- •GM-CSF is required for radiation-induced tumor reseeding."
Yes!
From the Vitalta M et. al. 2014 study: " ....We present evidence that irradiation of breast tumors can attract migrating breast cancer cells. Granulocyte-macrophage colony stimulating factor (GM-CSF) produced by tumor cells in response to radiation stimulates the recruitment of migrating tumor cells to irradiated tumors, suggesting a mechanism of tumor recurrence after radiation facilitated by transit of unirradiated, viable circulating tumor cells to irradiated tumors. Data supporting this hypothesis are presented through in vitro invasion assays and in vivo orthotopic models of breast cancer. Our work provides a mechanism for tumor recurrence in which radiation therapy attracts cells outside the radiation field to migrate to the site of treatment." Thus, irradiating breast tumors can actually attract non-irradiated circulating tumor cells back to the tumor for regrowth! (EDITORS' NOTE: Could this radiation factor into the proposed tumor seeding theory, in which breast cancer cells leave the tumor and can come back and re-seed the tumor? )
( For the study see Vitalta M et al., Recruitment of Circulating Breast Cancer Cells is Stimulated By Radiotherapy, Cell Rep, 2014.)
From the Vitalta M et. al. 2014 study: " ....We present evidence that irradiation of breast tumors can attract migrating breast cancer cells. Granulocyte-macrophage colony stimulating factor (GM-CSF) produced by tumor cells in response to radiation stimulates the recruitment of migrating tumor cells to irradiated tumors, suggesting a mechanism of tumor recurrence after radiation facilitated by transit of unirradiated, viable circulating tumor cells to irradiated tumors. Data supporting this hypothesis are presented through in vitro invasion assays and in vivo orthotopic models of breast cancer. Our work provides a mechanism for tumor recurrence in which radiation therapy attracts cells outside the radiation field to migrate to the site of treatment." Thus, irradiating breast tumors can actually attract non-irradiated circulating tumor cells back to the tumor for regrowth! (EDITORS' NOTE: Could this radiation factor into the proposed tumor seeding theory, in which breast cancer cells leave the tumor and can come back and re-seed the tumor? )
( For the study see Vitalta M et al., Recruitment of Circulating Breast Cancer Cells is Stimulated By Radiotherapy, Cell Rep, 2014.)
RADS MAY NOT ERADICATE ALL OF THE TUMOR
A 2015 STUDY HINTS AT ANOTHER REASON RADS MAY FAIL - RADS CAN LEAD TO TUMOR-INFILTRATING TREGS (T-CELLS), RESISTANCE, EVEN RESISTING FURTHER TREATMENT
*RADIATION THERAPY AND IMMUNE SYSTEM - CAN RADIATION INHIBIT - REALLY INTERFERE WITH - THE ANTI-CANCER IMMUNE RESPONSE?
YES! The Price J et al. 2015 study showed that, at least in the case of melanoma, radiation inhibits the anti-cancer response!
After radiation In skin tumors, Langerhans cells in the skin can resist apoptosis and make DNA repairs. An over-expressed checkpoint inhibitor leads to an increase in tumor-infiltrating regulatory T cells (Treg cells) . This immune response results in a failure to eradicate all the tumor cells. Radiation resistance!
How about in breast cancer?
The researchers note that there are dendritic cells, cousins of Langerhans cells, in other organs of the body!
YES! The Price J et al. 2015 study showed that, at least in the case of melanoma, radiation inhibits the anti-cancer response!
After radiation In skin tumors, Langerhans cells in the skin can resist apoptosis and make DNA repairs. An over-expressed checkpoint inhibitor leads to an increase in tumor-infiltrating regulatory T cells (Treg cells) . This immune response results in a failure to eradicate all the tumor cells. Radiation resistance!
How about in breast cancer?
The researchers note that there are dendritic cells, cousins of Langerhans cells, in other organs of the body!
The Price J et al. 2015 study found, at least in the case of melanoma, that radiation treatment "can lead to the accumulation of tumor-infiltrating regulatory T cells (Treg cells) and subsequent resistance of tumors to radiotherapy."
The study focused on epidermal mononuclear phagocytes Langerhans cells (LCs), "due to ability to resist depletion by high-dose ionizing radiation", and found that these "Langerhans cells resisted apoptosis and rapidly repaired DNA damage" after rads. Specifically, there is an over-expression of cyclin-dependent kinase inhibitor (CDKN1a) - a checkpoint inhibitor - that upregulates major histocompatibility complex class II molecules, which go to the lymph nodes and "induce an increase in Treg cell numbers" upon radiation.
Summary from Science Daily, 2015: "The team found that when radiotherapy damages skin harboring tumors, special skin immune cells called Langerhans cells are activated. These Langerhans cells can uniquely repair the damage in their own DNA caused by radiotherapy, allowing them to become resistant to radiotherapy and to even trigger an immune response causing skin tumors, such as melanoma, to resist further treatment. Investigators mimicked the effect of immunotherapy drugs called 'immune checkpoint inhibitors' to boost the immune system to attack tumors. This in turn blocked the ability of Langerhands cells to repair their own DNA after radiotherapy causing them to die, preventing an immune response that protects skin tumors."
Note that while skin tumors were the focus of this study, and on the Langerhans cells located in the skin, that radiation may fail to completely eradicate tumors in other organs of the body by this same process. "There cousins of Langerhans cells called dendritic cells are also activated by radiotherapy and the investigators stressed that it is critical we understand how they respond to treatment as well."
(For the study, see Price J et al.,CDKN1A Regulates Langerhans Cell Survival and Promotes Treg Cell Generation Upon Exposure to Ionizing Radiation, Nature Immunology, 2015. For the article, see Science Daily, How Immune Cells Resist Radiation, http://www.sciencedaily.com/releases/2015/11/151124170259.htm 2015,)
The study focused on epidermal mononuclear phagocytes Langerhans cells (LCs), "due to ability to resist depletion by high-dose ionizing radiation", and found that these "Langerhans cells resisted apoptosis and rapidly repaired DNA damage" after rads. Specifically, there is an over-expression of cyclin-dependent kinase inhibitor (CDKN1a) - a checkpoint inhibitor - that upregulates major histocompatibility complex class II molecules, which go to the lymph nodes and "induce an increase in Treg cell numbers" upon radiation.
Summary from Science Daily, 2015: "The team found that when radiotherapy damages skin harboring tumors, special skin immune cells called Langerhans cells are activated. These Langerhans cells can uniquely repair the damage in their own DNA caused by radiotherapy, allowing them to become resistant to radiotherapy and to even trigger an immune response causing skin tumors, such as melanoma, to resist further treatment. Investigators mimicked the effect of immunotherapy drugs called 'immune checkpoint inhibitors' to boost the immune system to attack tumors. This in turn blocked the ability of Langerhands cells to repair their own DNA after radiotherapy causing them to die, preventing an immune response that protects skin tumors."
Note that while skin tumors were the focus of this study, and on the Langerhans cells located in the skin, that radiation may fail to completely eradicate tumors in other organs of the body by this same process. "There cousins of Langerhans cells called dendritic cells are also activated by radiotherapy and the investigators stressed that it is critical we understand how they respond to treatment as well."
(For the study, see Price J et al.,CDKN1A Regulates Langerhans Cell Survival and Promotes Treg Cell Generation Upon Exposure to Ionizing Radiation, Nature Immunology, 2015. For the article, see Science Daily, How Immune Cells Resist Radiation, http://www.sciencedaily.com/releases/2015/11/151124170259.htm 2015,)
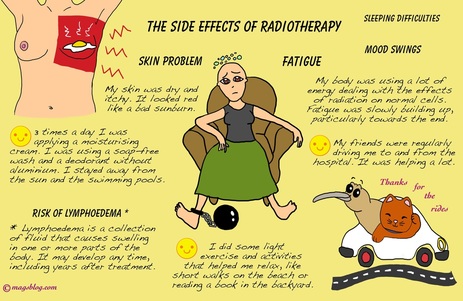
RADIATION THERAPY SIDE EFFECTS
We know that in a prospective study of breast cancer patients who had radiation therapy, "The group of patients that received radiation therapy only presented a more rapid decrease of total number of lymphocytes (part of your immune system) concerning the decrease observed in the group that underwent chemotherapy and radiation therapy."
(See Koyokoyrakis G et al, Post-Surgical Irradiation Causes Cellular Immune Suppression in Patients with Breast Cancer, Eur J cancer Care, 2009.)
(See Koyokoyrakis G et al, Post-Surgical Irradiation Causes Cellular Immune Suppression in Patients with Breast Cancer, Eur J cancer Care, 2009.)
SPECIFIC SKIN PROBLEMS FROM RADIATION THERAPY -EXCERPTS -www.webmed.com
External Beam Radiation
"Radiation passes through your skin, which can become red, swollen, warm, and sensitive -- as if you had a sunburn. It may peel or become moist and tender. Depending on the dose, you may lose hair or sweat less where you've been treated.
These skin reactions are common and short-term. They usually go away gradually within 4 to 6 weeks after your last treatment. Tell your doctor or nurse if you see skin changes outside the treated area.
Long-term side effects can last beyond a year after treatment. They may include a slight darkening of your skin, enlarged pores on your breast, more or less sensitive skin, thickening of breast tissue or skin, and a change in the size of the breast.
A rare complication of radiation is getting a new cancer or tumor where the radiation is given
Brachytherapy Radiation
Radioactive seeds or pellets as small as grains of rice are placed inside the breast, near the cancer. Whether this treatment might be right for you will depend on your tumor’s size, location, and other things.
The side effects include redness, bruising, breast pain, infection, weakness, and an increased risk of fractured ribs.
Brachytherapy can be used alone or with external beam radiation."
http://www.webmd.com/breast-cancer/breast-cancer-radiation-therapy-what-expect?page=2#1
External Beam Radiation
"Radiation passes through your skin, which can become red, swollen, warm, and sensitive -- as if you had a sunburn. It may peel or become moist and tender. Depending on the dose, you may lose hair or sweat less where you've been treated.
These skin reactions are common and short-term. They usually go away gradually within 4 to 6 weeks after your last treatment. Tell your doctor or nurse if you see skin changes outside the treated area.
Long-term side effects can last beyond a year after treatment. They may include a slight darkening of your skin, enlarged pores on your breast, more or less sensitive skin, thickening of breast tissue or skin, and a change in the size of the breast.
A rare complication of radiation is getting a new cancer or tumor where the radiation is given
Brachytherapy Radiation
Radioactive seeds or pellets as small as grains of rice are placed inside the breast, near the cancer. Whether this treatment might be right for you will depend on your tumor’s size, location, and other things.
The side effects include redness, bruising, breast pain, infection, weakness, and an increased risk of fractured ribs.
Brachytherapy can be used alone or with external beam radiation."
http://www.webmd.com/breast-cancer/breast-cancer-radiation-therapy-what-expect?page=2#1
HOW ABOUT HEART DAMAGE AFTER RADIATION THERAPY?
AT RISK FOR HEART DISEASE? RIGHT SIDE OR LEFT SIDE RADIATION? POSITION?
Highest cardiac risks after breast cancer radiation? Supine positioning during left-sided radiation.
Reducing risk? Prone positioning.
Reducing risk? Prone positioning.
In a 2014 study, Brenner DJ et al. stated, "Estimated lifetime risks of major coronary events for patients who receive radiotherapy for breast cancer are now in the range from 0.05% to 3.5%, with a typical value of 0.3% for a typical scenario."
Highest cardiac risks after radiation? Supine positioning during left-sided radiation.
Reducing risk? Prone positioning.
Right-sided radiation therapy? "For right-sided radiotherapy, where the heart is always out of field, cardiac doses and risks are smaller, and prone versus supine positioning has little effect, although prone position radiotherapy does reduce ipsilateral lung doses and thus reduces potential second lung cancer risks."
Multiplicative risks? For radiation exposure relative to cardiac disease, "the highest absolute radiation exposure risks correspond to the highest baselines cardiac risk (from smoking, etc.)"
(See Brenner DJ et al., Risk and Risk Reduction of major Coronary Events Associated with Contemporary Breast Radiotherapy, JAMA Internal Medicine, 2014.)
Why Does Radiation Cause Cardiac Abnormalities?
"There are several common pathways involved in the development of radiation-induced heart disease, including microvascular damage, inflammation, and fibrosis, although other pathways contribute. The interaction of multiple biochemical markers and cytokines, such as TGF-B and interleukins, drive a significant portion of chronic inflammation and late fibrosis."
Neil Taunk, 2015.
Neil Taunk, 2015.
In a 2015 review article, Neil Taunk writes that it is important to understand "the late effects of radiation to the chest. Radiation-induced heart disease comprises a spectrum of cardiac pathology including myocardial fibrosis and cardiomyopathy, coronary artery disease, valvular disease, pericardial disease, and arrhythmias. Tissue fibrosis is a common mediator in radiation-induced heart disease."
Indeed, "A major common endpoint for radiation-induced heart disease is tissue fibrosis. ...Radiation exposure leads to endothelial cell damage and subsequent microvasculature dysfunction due to fibrosis."
Further, "Radiation damage is characterized by both acute and chronic changes in cardiac tissue. Within minutes of ionizing radiation, cellular injury causes vasodilation and increased vascular permeability. Damaged endothelial cells secrete adhesion molecules and growth factors prompting activation of the acute inflammatory response. Recruited inflammatory cells secrete pro-fibrotic cytokines."
In conclusion: "There are several common pathways involved in the development of radiation-induced heart disease including microvascular damage, inflammation, and fibrosis, although other pathways contribute. The interaction of multiple biochemical markers and cytokines such as TGF-B and interleukins, drive a significant portion of chronic inflammation and late fibrosis."
(For the entire review article, see Neil Taunk, Radiation-Induced Heart Disease; Pathologic Abnormalities and Putative Mechanisms, Front Oncol, 2015.)
Indeed, "A major common endpoint for radiation-induced heart disease is tissue fibrosis. ...Radiation exposure leads to endothelial cell damage and subsequent microvasculature dysfunction due to fibrosis."
Further, "Radiation damage is characterized by both acute and chronic changes in cardiac tissue. Within minutes of ionizing radiation, cellular injury causes vasodilation and increased vascular permeability. Damaged endothelial cells secrete adhesion molecules and growth factors prompting activation of the acute inflammatory response. Recruited inflammatory cells secrete pro-fibrotic cytokines."
In conclusion: "There are several common pathways involved in the development of radiation-induced heart disease including microvascular damage, inflammation, and fibrosis, although other pathways contribute. The interaction of multiple biochemical markers and cytokines such as TGF-B and interleukins, drive a significant portion of chronic inflammation and late fibrosis."
(For the entire review article, see Neil Taunk, Radiation-Induced Heart Disease; Pathologic Abnormalities and Putative Mechanisms, Front Oncol, 2015.)
RADIATION AND MICROBIOME
RAT STUDY OF SINGLE OR MULTIPLE FRACTION TOTAL-BODY IRRADIATION SHOWS CHANGES IN MICROBIOME - EDITORS' NOTE: NOW WE NEED A STUDY ON BREAST CANCER RADIATION THERAPY AND THE MICROBIOME!!
Needing to rapidly determine if there has been heavy radiation damage from radiological terrorism, a nuclear power plant accident, or nuclear warfare, the Lam V et al team in their 2012 study, "hypothesized that intestinal microbiota excreted into the feces can act as a biomarker of radiation exposure".
For their experiment, rats received "single or multiple fraction total-body irradiation of 10.0 Gy and 18.0 Gy, respectively. " Fecal pellets were retrieved prior to treatment, then at days 4, 11, and 21 post-irradiation.
Results: "The radiation biomarkers consisted of increased 16S rRNA levels of 12 members of the Bacteroidales, Lactobacillaceae, and Streptocococcaceae after radiation exposure, unchanged levels of 98 Clostridiaceae and Peptostreptococcaceae, and decreased levels of 47 separate Clostridiaceae members; these biomarkers are present in human and rat feces."
Conclusion: "The present study has identified multiple intestinal microbiota present in feces that act as novel biomarkers of prior radiation exposure, where changes in expression are increased 100-1000-fold and where levels of reporting signals are detectable by 2 days post-exposure and persist for a minimum of three weeks after exposure."
EDITORS' NOTE: Whether or not we can extrapolate these high doses of total body irradiation in rats to any breast cancer radiation therapy, one wonders if there is a medical radiation accident? Will there be changes in the microbiota? Plus, we wonder if the microbiota may be disturbed by just any kind of breast cancer radiation therapy concomitant with scans.. Such a study is definitely needed.
(See Lam V et al., Intestinal Microbiota as Novel Biomarkers of Prior Radiation Exposure, Rad Res, 2012.)y