SURGERY
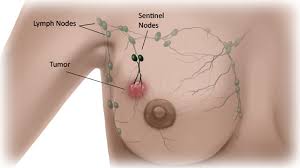
LYMPH NODES
AND
LYMPHEDEMA
LYMPH NODES
FEARFUL OF LYMPH NODE SURGERY?
Banished are the barbaric days when axilllary lymph node dissection, taking between 20-40 lymph nodes, was standard procedure to stage breast cancer. Axillary dissection can be physically disabling, leading to lymphedema.
Generally, now, a sentinel lymph node biopsy, entailing the taking of one or more lymph nodes draining from the tumor, is employed to stage the breast cancer. But, since breast cancer tumor cells can break away from the tumor and attach to a lymph vessel OR a blood vessel and travel around the body, a sentinel lymph node biopsy can not be definitive about breast cancer.
So, should you have a sentinel lymph node biopsy?
Arguments for removal .... A cancerous sentinel lymph node can potentially spread the cancer or, in the process of self-seeding, possibly re-seed the lymph node, or go back to the primary tumor.
Arguments against removal .... A sentinel lymph node biopsy still represents a loss of some of your precious immune system. Plus, there are always risks associated with surgery, such as spreading cancer cells. Will the information gleaned from the biopsy be helpful? What is your philosophy about radiation and/or chemo? ASCO 2014 guidelines state that for micrometastases or 1-2 cancerous lymph nodes, you do NOT need to have complete axillary dissection, so long as you have treatment (likely involving radiation, which could also increase lymphedema risk) to hopefully eradicate any residual cancer. In the event of a cancerous sentinel lymph node, the Cyr A et al. 2015 study suggests that "tumor biology" more than "tumor surgery" is a predictor of recurrence.... And don't forget the small (about 5%) increased risk of incurable lymphedema, and the life time attendant worries about blood being drawn, shots, bites etc. in the affected arm.
REMOVAL OF ANY LYMPH NODE IS ABHORRENT. BUT, ALTHOUGH VERY RARE, TWO MEMBERS OF THE BREAST CANCER STUDY AND SUPPORT GROUP DECLINED TO HAVE SWOLLEN, CANCEROUS LYMPH NODES REMOVED, AND THE LYMPH NODES GREW SO LARGE THAT THEY IMPINGED ON NERVES, AND THEY LOST THE USE OF THEIR ARMS. A THIRD PERSON, WHO ALSO REFUSED TO HAVE HER CANCEROUS LYMPH NODE REMOVED, SUFFERED INTERMITTENT PAIN.
READ THE MATERIAL HERE IN THE SURGERY-LYMPH NODES' SECTION, AS WELL AS OTHER RESEARCH, TO HELP MAKE UP YOUR OWN MIND ABOUT HAVING LYMPH NODE SURGERY!
Banished are the barbaric days when axilllary lymph node dissection, taking between 20-40 lymph nodes, was standard procedure to stage breast cancer. Axillary dissection can be physically disabling, leading to lymphedema.
Generally, now, a sentinel lymph node biopsy, entailing the taking of one or more lymph nodes draining from the tumor, is employed to stage the breast cancer. But, since breast cancer tumor cells can break away from the tumor and attach to a lymph vessel OR a blood vessel and travel around the body, a sentinel lymph node biopsy can not be definitive about breast cancer.
So, should you have a sentinel lymph node biopsy?
Arguments for removal .... A cancerous sentinel lymph node can potentially spread the cancer or, in the process of self-seeding, possibly re-seed the lymph node, or go back to the primary tumor.
Arguments against removal .... A sentinel lymph node biopsy still represents a loss of some of your precious immune system. Plus, there are always risks associated with surgery, such as spreading cancer cells. Will the information gleaned from the biopsy be helpful? What is your philosophy about radiation and/or chemo? ASCO 2014 guidelines state that for micrometastases or 1-2 cancerous lymph nodes, you do NOT need to have complete axillary dissection, so long as you have treatment (likely involving radiation, which could also increase lymphedema risk) to hopefully eradicate any residual cancer. In the event of a cancerous sentinel lymph node, the Cyr A et al. 2015 study suggests that "tumor biology" more than "tumor surgery" is a predictor of recurrence.... And don't forget the small (about 5%) increased risk of incurable lymphedema, and the life time attendant worries about blood being drawn, shots, bites etc. in the affected arm.
REMOVAL OF ANY LYMPH NODE IS ABHORRENT. BUT, ALTHOUGH VERY RARE, TWO MEMBERS OF THE BREAST CANCER STUDY AND SUPPORT GROUP DECLINED TO HAVE SWOLLEN, CANCEROUS LYMPH NODES REMOVED, AND THE LYMPH NODES GREW SO LARGE THAT THEY IMPINGED ON NERVES, AND THEY LOST THE USE OF THEIR ARMS. A THIRD PERSON, WHO ALSO REFUSED TO HAVE HER CANCEROUS LYMPH NODE REMOVED, SUFFERED INTERMITTENT PAIN.
READ THE MATERIAL HERE IN THE SURGERY-LYMPH NODES' SECTION, AS WELL AS OTHER RESEARCH, TO HELP MAKE UP YOUR OWN MIND ABOUT HAVING LYMPH NODE SURGERY!
2018 STUDY: IN LIMITED METASTASIS, LITTLE OR NO BENEFIT IN TAKING LYMPH NODES IN BREAST CANCER PATIENTS
"Indeed, several large, randomized clinical trials have demonstrated that ALN (axillary lymph node) resection for limited metastasis conferred minimal or no survival benefit for breast cancer patients."
EXCERPT FROM STUDY: "The role of ALN metastasis has been explained using a speedometer analogy. The authors assert that, just as removing the speedometer of a car does not reduce its speed, the removal of positive ALNs does not affect the rate of metastasis. In other words, ALN status is very useful for predicting the tumorigenic capability of the primary tumor but does not drive metastasis per se. Hence, dissection of positive ALNs will not reverse this capability, since spreading to distant sites appears to occur via a hematogenous (Note: By the blood), rather than a lymphatic, channel. Moreover, gene expression profiles of the primary tumor can predict metastasis location and survival, independent of ALN status, which further supports the idea that ALN metastasis is not the major factor in determining distant metastatic progression.. Finally, the genetic characterization of primary cancers provides important prognostic and predictive information that increasingly supersedes the informative capacity of lymph node status. Indeed, several large, randomized clinical trials have demonstrated that ALN resection for limited metastasis conferred minimal or no survival benefit for breast cancer patients."
(See Ullah I et al., Evolutionary History of Metastatic Breast Cancer Reveals Minimal Seeding from Axillary Lymph Nodes, J Clin Invest, 2018.)
"Indeed, several large, randomized clinical trials have demonstrated that ALN (axillary lymph node) resection for limited metastasis conferred minimal or no survival benefit for breast cancer patients."
EXCERPT FROM STUDY: "The role of ALN metastasis has been explained using a speedometer analogy. The authors assert that, just as removing the speedometer of a car does not reduce its speed, the removal of positive ALNs does not affect the rate of metastasis. In other words, ALN status is very useful for predicting the tumorigenic capability of the primary tumor but does not drive metastasis per se. Hence, dissection of positive ALNs will not reverse this capability, since spreading to distant sites appears to occur via a hematogenous (Note: By the blood), rather than a lymphatic, channel. Moreover, gene expression profiles of the primary tumor can predict metastasis location and survival, independent of ALN status, which further supports the idea that ALN metastasis is not the major factor in determining distant metastatic progression.. Finally, the genetic characterization of primary cancers provides important prognostic and predictive information that increasingly supersedes the informative capacity of lymph node status. Indeed, several large, randomized clinical trials have demonstrated that ALN resection for limited metastasis conferred minimal or no survival benefit for breast cancer patients."
(See Ullah I et al., Evolutionary History of Metastatic Breast Cancer Reveals Minimal Seeding from Axillary Lymph Nodes, J Clin Invest, 2018.)
Lymph nodes are immune organs.
Whether or not you decide to have a sentinel lymph
node biopsy, or any other additional lymph nodes taken,
please be aware that tumor draining lymph nodes
may be sick immune organs. You need to boost your immune
system. Some potential immune problems of tumor draining
lymph nodes are highlighted in the box below, with more
discussion to follow in this section.
"Within tumor- draining lymph nodes, tumor invasion is only distinguished between isolated tumor cells (<0.2 mm), micrometastasis (0.2–2 mm), and metastasis (>2 mm). Even though lymph nodes are immune organs, changes in immune cell populations, such as cell numbers, densities, and spatial relationships, are often unexplored." Setiadi A et al., 2010.
Whether or not you decide to have a sentinel lymph
node biopsy, or any other additional lymph nodes taken,
please be aware that tumor draining lymph nodes
may be sick immune organs. You need to boost your immune
system. Some potential immune problems of tumor draining
lymph nodes are highlighted in the box below, with more
discussion to follow in this section.
"Within tumor- draining lymph nodes, tumor invasion is only distinguished between isolated tumor cells (<0.2 mm), micrometastasis (0.2–2 mm), and metastasis (>2 mm). Even though lymph nodes are immune organs, changes in immune cell populations, such as cell numbers, densities, and spatial relationships, are often unexplored." Setiadi A et al., 2010.
THE IMPORT OF A SWOLLEN, CANCEROUS LYMPH NODE?
CANCEROUS TUMOR DRAINING LYMPH NODES ARE
SICK IMMUNE ORGANS.
* " ...Even tumor free axillary lymph nodes exhibited changes in immune profile..." Khort H et al., 2005
* "The spatial grouping of T and B cells differed between healthy and breast cancer lymph nodes, and this could be attributed to the lack of B cell localization in the extrafollicular region (where there is immune surveillance) of the tumor draining lymph nodes." Setiadi A et al., 2010.
* "Dendritic cells (process antigens from invaders) in healthy lymph nodes aggregate in large clusters of mature cells, whereas dendritic cells in tumor draining lymph nodes tend to remain unclustered or form clusters with fewer mature cells (impairing anti-tumor response)." Chang A et al., 2013.
* "The extra-cellular matrix composition and, in particular, the collagen fiber density, of lymph nodes, may be an informative parameter for assessing the aggressiveness of a particular breast cancer." Rizman A et al., 2015.
CANCEROUS TUMOR DRAINING LYMPH NODES ARE
SICK IMMUNE ORGANS.
* " ...Even tumor free axillary lymph nodes exhibited changes in immune profile..." Khort H et al., 2005
* "The spatial grouping of T and B cells differed between healthy and breast cancer lymph nodes, and this could be attributed to the lack of B cell localization in the extrafollicular region (where there is immune surveillance) of the tumor draining lymph nodes." Setiadi A et al., 2010.
* "Dendritic cells (process antigens from invaders) in healthy lymph nodes aggregate in large clusters of mature cells, whereas dendritic cells in tumor draining lymph nodes tend to remain unclustered or form clusters with fewer mature cells (impairing anti-tumor response)." Chang A et al., 2013.
* "The extra-cellular matrix composition and, in particular, the collagen fiber density, of lymph nodes, may be an informative parameter for assessing the aggressiveness of a particular breast cancer." Rizman A et al., 2015.
LYMPH NODES' BACKGROUND - DEFINTION
"A lymph node is an oval-shaped organ of the lymphatic system, distributed widely throughout the body including the armpit and stomach and linked by lymphatic vessels. Lymph nodes are major sites of B, T, and other immune cells. Lymph nodes are important for the proper functioning of the immune system, acting as filters for foreign particles and cancer cells." (See wikipedia.org.)
WHICH BREAST CANCER TYPES HAVE MORE POSITIVE LYMPH NODES?
TRIPLE POSITIVE- ER+, PR+, HER2+ - MOST POSITIVE LYMPH NODES
TRIPLE NEGATIVE - ER- , PR-, HER2- - LEAST POSITIVE LYMPH NODES
In the Chengshuai S et al 2014 study, the researchers sought to predict the lymph node status, that is, which lymph nodes will test positive for cancer, according to the subtype of breast cancer.
"Predictors used to identify the axillary node status has been studied before . Tumor size is the most valuable predictor for axillary node status in patients with breast cancer ... Besides, potential predictors include lymphavascular invasion , age at diagnosis, and so on."
Employing a database, out of selected 814 breast cancer patients, "The number and the percentage (in parentheses) of each type of breast cancer is as follows: Luminal A 230 (28.3%), Luminal Her2- 284 (34.9%), Luminal Her2+ 104 (12.8%), HER2+ 72 (8.8%), triple negative breast cancer 124 (15.2%). ... Tumor size and tumor subtype show statistical significance with lymph node involvement. Using triple negative as a reference, both Luminal B type (Luminal HER2-, Luminal HER2+) shows significant higher probability of lymph node involvement."
Conclusion: " ... Lymph node involvement is an intrinsic characteristic for molecular subtype of breast cancer. Triple positive and triple negative breast cancer accounts the most and least possibility of lymph node involvement." (See Chengshuai S et al., Association Between Molecular Subtypes and Lymph Node Status in Invasive Breast Cancer, Int J Clin Exp Pathol, 2014.)
POSITIVE LYMPH NODES REFLECT BREAKDOWN OF IMMUNE SYSTEM
NODE-POSITIVE BREAST CANCER HAS BREAKDOWN OF IMMUNE RESPONSE IN TUMOR, BLOOD, AND TUMOR-DRAINING LYMPH NODES
"Overall, significant differences were found in immune cell gene expression signatures from tumor-draining lymph nodes between node-negative and node-positive breast cancer patients. In comparison to node-negative patients, node-positive patients show significant down regulation of immune-related pathways such as antigen presentation, and up regulation of cell cycle and tumor-promoting pathways. Similar patterns were also observed systemically in the peripheral blood compartment and to some extent also in the primary tumor compartment."
Zuckerman N et al., 2014
Zuckerman N et al., 2014
Tumor cells disseminate early, and metastasis may have started 5-7 years before a diagnosis of breast cancer.
So why do only some people have lymph nodes that are positive for cancer?
This is precisely what the Zuckerman group wanted to know.
In their 2014 study, Zuckerman N et al. "hypothesized that the host immune response in node- negative patients is more intact and thereby can resist tumor invasion compared to node- positive patients. As such, lymph node metastasis requires breakdown of the host immune response in addition to the escape of cancer cells from the tumor."
The researchers investigated the areas where cancer and immune cells interact: TUMOR, TUMOR-DRAINING LYMPH NODES, AND PERIPHERAL BLOOD.
Findings: "Overall, significant differences were found in purified immune cell gene expression signatures from tumor-draining lymph nodes between node-negative and node-positive breast cancer patients. In comparison to node-negative patients, node-positive patients show significant down regulation of immune-related pathways such as antigen presentation, and up regulation of cell cycle and tumor-promoting pathways. Similar patterns were also observed systemically in the peripheral blood compartment and to some extent also in the primary tumor compartment."
Conclusion: While tumor cells can potentially go to the lymph nodes, the lymph nodes, as immune organs, should be able to "resist tumor invasion". Unfortunately, not so. Why not? How is it that the node-negative breast cancer patient has been able to fend off tumor invasion? The researchers' findings "suggest that the key difference lies in node-negative patients' immune responses being more intact and thus capable of responding to and eradicating tumors metastases in tumor-draining lymph nodes, while node-positive patients' immune cells are modulated to other, tumor-promoting processes. ....These differences, which are shown to be local and systemic using various bioinformatics methods, suggest that node-positive patients’ immune-related signatures are down-regulated, and tumor-promoting signatures are up-regulated compared to node-negative patients."
(See Zuckerman N et al., Altered Local and Systemic Immune Profiles Underlie Lymph Node Metastasis in Breast Cancer Patients, Int J Cancer, 2013.)
BUT DO YOU HAVE ANY POSITIVE LYMPH NODES? TESTING WITH A SENTINEL LYMPH NODE BIOPSY - DEFINITION
From www.cancer.gov:
How breast cancer cells spread:: "Breast cancer cells are most likely to spread first to lymph nodes located in the axilla, or armpit area, next to the affected breast. However, in breast cancers close to the center of the chest (near the breastbone), cancer cells may spread first to lymph nodes inside the chest (under the breastbone) before they can be detected in the axilla.The number of lymph nodes in the axilla varies from person to person but usually ranges from 20 to 40. Historically, removal of these lymph nodes (in an operation called axillary lymph node dissection) was done for two reasons: to help stage breast cancer and to help prevent a regional recurrence of the disease. (Regional recurrence of breast cancer occurs when breast cancer cells that have migrated to nearby lymph nodes give rise to a new tumor.) Because removing multiple lymph nodes at the same time has been associated with adverse effects, the possibility that sentinel lymph node biopsy alone might be sufficient for staging breast cancer in women who have no clinical signs of axillary lymph node metastasis, such as swollen or “matted” (clumped or stuck together) nodes, was investigated." (See
http://www.cancer.gov/about-cancer/diagnosis-staging/staging/sentinel-node-biopsy-fact-sheet#q8.)
What is a sentinel lymph node biopsy? During the sentinel lymph node biopsy, "the sentinel lymph node is identified, removed, and examined to determine whether cancer cells are present.A negative sentinel lymph node result suggests that cancer has not developed the ability to spread to nearby lymph nodes or other organs. A positive sentinel lymph node result indicates that cancer is present in the sentinel lymph node and may be present in other nearby lymph nodes (called regional lymph nodes) and, possibly, other organs. This information can help a doctor determine the stage of the cancer (extent of the disease within the body) and develop an appropriate treatment plan.
How is the sentinel lymph node biopsy conducted? "A surgeon injects a radioactive substance, a blue dye, or both near the tumor to locate the position of the sentinel lymph node. The surgeon then uses a device that detects radioactivity to find the sentinel node or looks for lymph nodes that are stained with the blue dye. Once the sentinel lymph node is located, the surgeon makes a small incision (about 1/2 inch) in the overlying skin and removes the node." The sentinel lymph node biopsy can be done at the time of surgery for the primary tumor, or before or after it.. See http://www.cancer.gov/about-cancer/diagnosis-staging/staging/sentinel-node-biopsy-fact-sheet#q4.
How breast cancer cells spread:: "Breast cancer cells are most likely to spread first to lymph nodes located in the axilla, or armpit area, next to the affected breast. However, in breast cancers close to the center of the chest (near the breastbone), cancer cells may spread first to lymph nodes inside the chest (under the breastbone) before they can be detected in the axilla.The number of lymph nodes in the axilla varies from person to person but usually ranges from 20 to 40. Historically, removal of these lymph nodes (in an operation called axillary lymph node dissection) was done for two reasons: to help stage breast cancer and to help prevent a regional recurrence of the disease. (Regional recurrence of breast cancer occurs when breast cancer cells that have migrated to nearby lymph nodes give rise to a new tumor.) Because removing multiple lymph nodes at the same time has been associated with adverse effects, the possibility that sentinel lymph node biopsy alone might be sufficient for staging breast cancer in women who have no clinical signs of axillary lymph node metastasis, such as swollen or “matted” (clumped or stuck together) nodes, was investigated." (See
http://www.cancer.gov/about-cancer/diagnosis-staging/staging/sentinel-node-biopsy-fact-sheet#q8.)
What is a sentinel lymph node biopsy? During the sentinel lymph node biopsy, "the sentinel lymph node is identified, removed, and examined to determine whether cancer cells are present.A negative sentinel lymph node result suggests that cancer has not developed the ability to spread to nearby lymph nodes or other organs. A positive sentinel lymph node result indicates that cancer is present in the sentinel lymph node and may be present in other nearby lymph nodes (called regional lymph nodes) and, possibly, other organs. This information can help a doctor determine the stage of the cancer (extent of the disease within the body) and develop an appropriate treatment plan.
How is the sentinel lymph node biopsy conducted? "A surgeon injects a radioactive substance, a blue dye, or both near the tumor to locate the position of the sentinel lymph node. The surgeon then uses a device that detects radioactivity to find the sentinel node or looks for lymph nodes that are stained with the blue dye. Once the sentinel lymph node is located, the surgeon makes a small incision (about 1/2 inch) in the overlying skin and removes the node." The sentinel lymph node biopsy can be done at the time of surgery for the primary tumor, or before or after it.. See http://www.cancer.gov/about-cancer/diagnosis-staging/staging/sentinel-node-biopsy-fact-sheet#q4.
WHAT IF A RADIOACTIVE SUBSTANCE IS USED DURING THE SENTINEL LYMPH NODE BIOPSY?
(EDITORS' NOTES: Likely, a radioactive tracer, technetium sulfur colloid, will be used in your sentinel lymph node procedure. But do be aware that if a radioactive tracer is employed, you may be exposed to some radiation. Let's just hope that the radiation has been minimalized to protect the patients, the surgical staff, and the pathology staff since the 2010 study cited here.)
In the Renshaw A et al. 2010 study, pathologists, who have to handle surgical specimens, evaluated the radiation emanating from sentinel node biopsies and other resections to see how much radiation they are being exposed to " During the period studied (from 2003 - 2009), 2,902 sentinel node and primary resection specimens were received in our pathology laboratory.. All badges worn by pathology assistants and histotechnologists were always less than 10 mrem/mo. The percentage of specimens with greater than background radiation (≥0.2 mrem/h) rose from 6.3% to 34.8% from 2003 to 2009 ; specimens with more than 10 mrem/h ro
(See Renshaw A et al., Increasing Radiation from Sentinel Node Specimens in Pathology Over Time, American Journal of Clinical Pathology, 2010.)
(EDITORS' NOTES: Likely, a radioactive tracer, technetium sulfur colloid, will be used in your sentinel lymph node procedure. But do be aware that if a radioactive tracer is employed, you may be exposed to some radiation. Let's just hope that the radiation has been minimalized to protect the patients, the surgical staff, and the pathology staff since the 2010 study cited here.)
In the Renshaw A et al. 2010 study, pathologists, who have to handle surgical specimens, evaluated the radiation emanating from sentinel node biopsies and other resections to see how much radiation they are being exposed to " During the period studied (from 2003 - 2009), 2,902 sentinel node and primary resection specimens were received in our pathology laboratory.. All badges worn by pathology assistants and histotechnologists were always less than 10 mrem/mo. The percentage of specimens with greater than background radiation (≥0.2 mrem/h) rose from 6.3% to 34.8% from 2003 to 2009 ; specimens with more than 10 mrem/h ro
(See Renshaw A et al., Increasing Radiation from Sentinel Node Specimens in Pathology Over Time, American Journal of Clinical Pathology, 2010.)
SENTINEL LYMPH NODE MORBIDITY (SUCH AS PAIN, SWELLING)? - MINIMAL MORBIDITY
The Alsaif AA et al. 2015 study evaluated sentinel lymph node biopsies in breast cancer. 95 breast cancer patients who were sentinel lymph node negative had no immediate axillary dissection. Further testing revealed that ten of those patients had micrometastases, and six of those patients had an axillary dissection. After a median follow-up of 35.5 months, there was only one axillary recurrence. Morbidity was minimal. Thus, "The obtainable results from our local experience in SLNB in breast cancer, concur with that seen in published similar literature in particular the axillary failure rate. Sentinel lymph node biopsy resulted in minimal morbidity."
(See Alsaif AA et al., Sentinel Lymph Node Biopsy in Breast Cancer, Saudi Med J, 2015.)
YOU HAVE HAD A SENTINEL LYMPH NODE BIOPSY. WHAT NOW? - 2014 LYMPH NODE SURGERY (ASCO) GUIDELINE UPDATES
"1. Women without sentinel node metastases should not receive axillary lymph node dissection.
2. Most women with 1 to 2 metastatic sentinel lymph nodes planning to receive breast conserving surgery with whole breast radiotherapy should not undergo axillary lymph node dissection.
3. Women with sentinel lymph node metastases who will receive mastectomy may be offered axillary lymph node dissection."
(See http://www.asco.org/press-center/asco-updates-guideline-sentinel-node-biopsy-early-stage-breast-cancer-evidence-supports.)
DIAGNOSED WITH MICROMETASTES IN LYMPH NODES? - GOOD DIAGNOSIS WITH TINY, TINY METASTASES - MICROMETASTASES? ISOLATED TUMOR CELLS? DON'T WANT AN AXILLARY DISSECTION?
FINE! SAME SURVIVAL RATE AS THOSE HAVING AXILLARY DISSECTION!
"In this nationwide study, we found a low risk of axillary recurrences of 1.58 % and we did not find a significantly increased risk of axillary recurrences if axillary dissection was omitted in patients with micrometastases or isolated tumor cells in sentinel nodes. Furthermore, no significant difference in overall survival was seen between patients with and without axillary dissection when adjusting for adjuvant treatment."
Tvedshov TF et al., 2015
Tvedshov TF et al., 2015
Up to 23% of breast cancer patients are reported to have either micrometastases of isolated tumor cells.
According to the AJCC/UICC staging manuals (2002), micrometastases are defined "to be >0.2 mm but not >2.0 mm and introduced the term isolated tumor cells that are ≤0.2 mm".(Medscape,http://www.medscape.com/viewarticle/770016_4.)
In the Tvedshov TF et al. 2015 study, the researchers "estimated the impact of axillary lymph node dissection on the risk of axillary recurrence and overall survival in breast cancer patients with micrometastases or isolated tumor cells in sentinel nodes".
In this study, the researchers evaluated 2074 patients who had either micrometastases or isolated tumor cells, of which 240 did not undergo further axillary surgery. "The 5-year cumulated incidence for axillary recurrence was 1.58 %. No significant difference in axillary recurrences were seen between patients with and without axillary node dissection."
Summary: "There was no significant difference in overall survival between patients with and without axillary node dissection, when adjusting for age, co-morbidity, tumor size, histology type, malignancy grade, lymphovascular invasion, hormone receptor status, adjuvant systemic treatment and radiotherapy, with a hazard ratio for death if axilalry dissection was omitted of 1.21 in patients with micrometastases and 0.96 in patients with isolated tumor cells after a medium follow-up on 8 and 5 years. In this nationwide study, we found a low risk of axillary recurrences of 1.58 % and we did not find a significantly increased risk of axillary recurrences if axillary dissection was omitted in patients with micrometastases or isolated tumor cells in sentinel nodes. Furthermore, no significant difference in overall survival was seen between patients with and without axillary dissection when adjusting for adjuvant treatment."
(See Tvedskoc TF et al., Prognostic Significance of Axillary Dissection in Breast Cancer Patients with Micrometastases or Isolated Tumor Cells in Sentinel Nodes: A Nationwide Study, Breast Cancer Res Treat, 2015.)
DIAGNOSED WITH MICROMETASTES IN LYMPH NODES? GOOD DIAGNOSIS WITH TINY, TINY METASTASES - NO NEED FOR SYSTEMIC THERAPY!
In the Gobhardan P et al. 2011 study, the researchers evaluated micrometastases in sentinel lymph node biopsies.
Conclusion: The researchers found that there was no difference in disease- free survival or overall survival in people with micrometastases. "The present data from a large multi-center population with a long-term follow-up (around 6.4 years) do not support the use of adjuvant system treatment in patients only because they have micrometastases."
(See Gobardhan P et al., Prognostic Value of Lymph Node Micrometastases in Breast Cancer: A Multi-Center Cohort Study, Breast Oncology, 2011.)
DIAGNOSED WITH A POSITIVE SENTINEL LYMPH NODE? - ASCO -- SKIP AXILLARY DISSECTION IF 1-2 POSITIVE SENTINEL LYMPH NODES + THERAPY
REVIEW STUDY BELOW -- IT IS NOT SURGERY, BUT THE BIOLOGY (ESTROGEN RECEPTOR NEGATIVITY AND LYMPHOVASCULAR INVASION) THAT PREDICT RECURRENCE.
This study "supports the idea that axillary dissection is of limited value for local control. In fact, women in whom axillary dissection was omitted were not significantly more likely to experience recurrence at any site and did not have higher disease-specific mortality. Increased recurrence risk is instead seen with higher grade, ER and PR negativity, and lymphovascular invasion, suggesting that tumor biology more than surgical therapy is a predictor of recurrence."
Cyr A et al., 2012
Cyr A et al., 2012
In the Cyr A et al. 2012 study, the researchers found that axillary dissection in breast cancer patients with a positive sentinel lymph node may be safely omitted.
In their study, "Women with a positive sentinel lymph node diagnosed between 1999 and 2010 were included in this review and were stratified according to whether they did or did not undergo complete axillary lymph node dissection. Primary endpoints included recurrence and breast cancer-specific mortality."
Results: "This study demonstrates no axillary recurrences at a median follow-up of 73 months in women with a positive sentinel node who did not undergo axillary dissection. Our findings are similar to those of other studies reporting low axillary recurrence rates when axillary dissection is omitted despite the presence of sentinel node disease." ... Other studies and our study "support the idea that axillary dissection is of limited value for local control. In fact, women in whom axillary dissection was omitted were not significantly more likely to experience recurrence at any site and did not have higher disease-specific mortality. Increased recurrence risk is instead seen with higher grade, ER and PR negativity, and lymphovascular invasion, suggesting that tumor biology more than surgical therapy is a predictor of recurrence."
Conclusion: "Omission of axillary node dissection in women with sentinel node disease does not significantly impact in-breast, nodal, or distant recurrence or mortality."
(See Cyr A et al., Disease Recurrence in Sentinel Node-Positive Breast Cancer Patients Forgoing Axillary Lymph Node Dissection, Ann Surg Oncol, 2012.)
YOU HAVE POSITIVE LYMPH NODES - WHAT DOES THIS MEAN?
T -CELL AND B- CELL DIFFERENCES
LOCATION, LOCATION, LOCATION!!
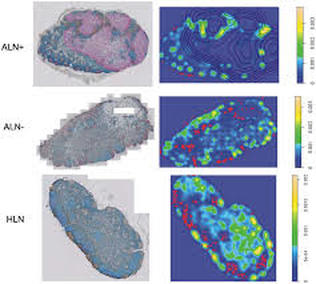
Figure 5. Representative whole-section images and density plots of B cells in an ALN+, ALN- and HLN. The proportion of B cell isolates is the highest in the HLNs. On whole-section images: blue: T cells; brown: B cells; red: tumor cells.
On density plots: red dots: B cell isolates (lowest 15% of density); yellow and green: B cell aggregates (highest 85% of density); lines: tumor contours. (Note: HLN are healthy lymph nodes, ALN are axillary lymph nodes, either positive [+] or negative [-].)
Comparison of the percentages of lymph nodes found with matching spatial grouping patterns of T and B cells and the average proportion of B cell isolates in tumor-free regions.
Axillary Lymph Nodes + 10%
Axillary Lymph Nodes - 40%
Sentinel Node Positive 47%
Health Lymph Nodes 57%
On density plots: red dots: B cell isolates (lowest 15% of density); yellow and green: B cell aggregates (highest 85% of density); lines: tumor contours. (Note: HLN are healthy lymph nodes, ALN are axillary lymph nodes, either positive [+] or negative [-].)
Comparison of the percentages of lymph nodes found with matching spatial grouping patterns of T and B cells and the average proportion of B cell isolates in tumor-free regions.
Axillary Lymph Nodes + 10%
Axillary Lymph Nodes - 40%
Sentinel Node Positive 47%
Health Lymph Nodes 57%
Within tumor-draining lymph nodes, "tumor invasion is only distinguished between isolated tumor cells (<0.2 mm), micrometastasis (0.2–2 mm), and metastasis (>2 mm)".
Okay, but what about changes in the immune cell populations in the cancerous lymph nodes? Cell number densities? Spatial relationships?
These changes are "often unexplored".
In the Setiadi A et al. 2010 study, the researchers set about to assay the immune cells in both cancerous and cancer-free lymph nodes, using a "novel quantitative imaging analysis approach" to study the "architectural patterns of T and B cells within tumor-draining lymph nodes from breast cancer patients versus healthy lymph nodes".
Background: "Upon entry from the bloodstream, T and B cells home to different regions in lymph nodes. Prior to follicular (a compartment in the lymph node) homing and development of antibody responses, extrafollicular (outside the follicle) localization of B cells is an important event that allows B cells to survey local antigen-carrying antigen-presenting cells (these are cells that present an antigen, which is some foreign toxin) and interact with T-helper cells. Reduction of B cells in the extrafollicular T cell zones may lead to ineffective interaction with antigen presenting cells and impairment of T cell-dependent B cells activation, which can ultimately contribute to defective anti-tumor immune responses."
Findings: In examining the T and B cells, the researchers "found that even when the proportions of T and B cells are similar, the spatial grouping patterns of these cells differed between healthy and tumor-draining lymph nodes".
Conclusion: "We found that the spatial grouping patterns of T and B cells differed between healthy and breast cancer lymph nodes, and this could be attributed to the lack of B cell localization in the extrafollicular region of the tumor-draining lymph nodes."
(See Setiadi A et al., Quantitative, Architectural Analysis of Immune Cell Subsets in Tumor-Draining Lymph Nodes from Breast Cancer Patients and Healthy Lymph Nodes, PLOS, 2010.)
Okay, but what about changes in the immune cell populations in the cancerous lymph nodes? Cell number densities? Spatial relationships?
These changes are "often unexplored".
In the Setiadi A et al. 2010 study, the researchers set about to assay the immune cells in both cancerous and cancer-free lymph nodes, using a "novel quantitative imaging analysis approach" to study the "architectural patterns of T and B cells within tumor-draining lymph nodes from breast cancer patients versus healthy lymph nodes".
Background: "Upon entry from the bloodstream, T and B cells home to different regions in lymph nodes. Prior to follicular (a compartment in the lymph node) homing and development of antibody responses, extrafollicular (outside the follicle) localization of B cells is an important event that allows B cells to survey local antigen-carrying antigen-presenting cells (these are cells that present an antigen, which is some foreign toxin) and interact with T-helper cells. Reduction of B cells in the extrafollicular T cell zones may lead to ineffective interaction with antigen presenting cells and impairment of T cell-dependent B cells activation, which can ultimately contribute to defective anti-tumor immune responses."
Findings: In examining the T and B cells, the researchers "found that even when the proportions of T and B cells are similar, the spatial grouping patterns of these cells differed between healthy and tumor-draining lymph nodes".
Conclusion: "We found that the spatial grouping patterns of T and B cells differed between healthy and breast cancer lymph nodes, and this could be attributed to the lack of B cell localization in the extrafollicular region of the tumor-draining lymph nodes."
(See Setiadi A et al., Quantitative, Architectural Analysis of Immune Cell Subsets in Tumor-Draining Lymph Nodes from Breast Cancer Patients and Healthy Lymph Nodes, PLOS, 2010.)
YOU HAVE POSITIVE LYMPH NODES. WHAT DOES THIS MEAN?- DENDRITIC CELL ANTI-TUMOR LYMPHOCYTE ACTIVATION DIFFERENCES
CLUSTERS, CLUSTERS, CLUSTERS
MATURITY, MATURITY, MATURITY
Immune cells not just by numbers, but by spatial organization.
In the Chang A et al. 2013 study, the researchers sought to characterize not only the numerical changes in the immune cells that are known to be "significantly decreased" in tumor-draining lymph nodes, but how the spatial organization of specific dendritic cells may have an impact on a breast cancer patient's prognosis.
What are dendritic cells? "An anti-tumor response is elicited when dendritic cells present tumor antigens (toxins or foreign substances) to T cells leading to activation and proliferation of cancer-specific T cells."
In this study, the researchers "analyzed immune cells within tumor-draining lymph nodes from 59 breast cancer patients with at least 5 years of clinical follow-up using immunohistochemical staining with a novel quantitative image analysis system. We developed algorithms to analyze spatial distribution patterns of immune cells in cancer versus healthy intra-mammary lymph nodes to derive information about possible mechanisms underlying immune-dysregulation in breast cancer."
Findings: Reduced CD83 mature dendritic cells, less lymphocyte activation in metastatic lymph nodes. Specifically, "The degree of clustering of dendritic cells (in terms of spatial proximity of the cells to each other) was reduced in tumor-draining lymph nodes compared to healthy lymph nodes." Dendritic cell clusters in tumor-draining lymph nodes were more numerous than in healthy lymph nodes, but there were actually fewer dendritic cells, as well as fewer dendritic cells bearing the hallmark maturity marker, CD83. T cells increase around dendritic clusters, but were found to be reduced in the tumor-draining lymph nodes as compared to the healthy lymph nodes. "Importantly, clinical outcome analysis revealed that dendritic cell clustering in cancerous tumor-draining lymph nodes correlated with the duration of disease-free survival in breast cancer patients."
Conclusion: "A novel hypothesis of this study is that not only numerical changes, but also spatial organization of dendritic cells may impact immune function and clinical outcome in cancer. Using novel spatial analysis algorithms, we found that dendritic cells in healthy lymph nodes aggregate into large clusters, predominantly composed of mature dendritic cells. ...To our knowledge, this is the first report to demonstrate that the spatial organization of dendritic cells within tumor-draining lymph nodes impact clinical outcome in human cancer."
(See Chang A et al., Spatial Organization of Dendritic Cells Within Tumor Draining Lymph Nodes Impacts Clinical Outcome in Breast Cancer Patients, Journal of Translational Medicine, 2013.)
YOU HAVE POSITIVE LYMPH NODES. WHAT DOES THIS MEAN? - METASTATIC XENOGRAFTS IN MICE INCREASE COLLAGEN FIBER DENSITY IN LYMPH NODES WHICH MAY PORTEND AGGRESSIVENESS OF BREAST CANCER
COLLAGEN DENSITY, COLLAGEN DENSITY, COLLAGEN DENSITY
"We have shown here that the ECM (extracellular matrix) composition, and in particular the collagen fiber density, of lymph nodes may be an informative parameter for assessing the aggressiveness of a given breast cancer, which could be measured as part of the pathology work-up of the removed lymph nodes."
"Collagen fibers are an important component of the extracellular matrix (secreted extracellular molecules that help with structure and sustenance of the cells), and increased stromal collagen in primary tumors can facilitate breast tumor formation, invasion, and metastasis."
How about cancerous lymph nodes?
Can cancer cells that have metastasized to the lymph nodes modify the extracellular matrix, which includes collagen?
Yes!
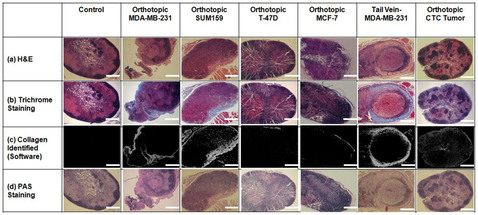
In the Rizwan A et al. 2015 study, the researchers investigated extracellular matrix molecules in breast cancer xenografts in mice.
Results: "In this study, we have identified distinct changes in the extracellular components of lymph nodes, in particular in their collagen fiber matrix, in response to the presence of metastatic breast cancer cells in the lymph nodes of mouse models of breast cancer. Sentinel lymph nodes in mice growing metastatic human breast tumor xenografts, or growing xenografts generated from circulating tumor cells, exhibited increased collagen density and increased periodic acid – Schiff (PAS) positive basement membrane, polysaccharides, and mucosubstances." Note, specifically, that metastatic estrogen-negative xenografts in the mice and tumor xenografts from circulating tumor cells in the mice "displayed an increased collagen I density" compared to mice without any tumors and mice with non-metastatic estrogen-positive tumor xenografts.
Conclusion: The researchers have demonstrated " that the extracellular matrix composition, and in particular the collagen fiber density, of lymph nodes may be an informative parameter for assessing the aggressiveness of a given breast cancer, which could be measured as part of the pathology work-up of the removed lymph nodes.."
(See Rizwan A et al., Metastatic Breast Cancer Cells in Lymph Nodes Increase Nodal Collagen Density. Scientific Reports, 2015.
"Epidemiological analyses of breast cancer patients indicated that metastasis might be initiated already 5–7 years before diagnosis of the primary tumor, yet only a subset of patients develop clinically evident relapse. Under this new view, the host immune response plays an important role in suppressing micrometastases and determines if patients develop clinically evident metastasis in lymph nodes or distant organs.."
Zuckerman N et al., 2013..
Zuckerman N et al., 2013..
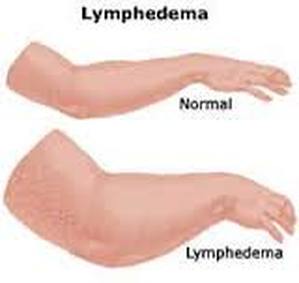
LYMPHEDEMA
2007 STUDY SHOWED UNDETECTED LYMPH CHANNELS CONNECTING SUPERFICIAL AND DEEP LYMPHATIC SYSTEM THAT HAD OPENED UP IN ARM WITH INCOMPLETE AXILLARY DISSECTION
LYMPHEDEMA PREVENTED!!
THE EDITORS ARE SMITTEN WITH THIS STUDY!! SUCH A HOPEFUL STUDY!
In the Suami H et al. 2007 study, the researchers' objective was to determine the differences in lymph structure in the upper arm following an axillary dissection.
To this end, the researchers compared a normal arm to an arm that had undergone an axillary dissection in the cadaver of an 81 year old woman..
Results: "...The mastectomy side showed remarkable differences and revealed that the lymph node clearance in the axilla had been incomplete on that side." As expected, there was an "obliteration of superficial lymph vessels". Also, there was "interval lymph node enlargement" in the arm. "The major difference was the almost complete absence of the superficial lymphatic network in the right arm, proximal to the elbow, because of fibrosis and blockage of the lymphatic channels. A circuitous pathway was identified that bypassed the blocked lymphatics in the arm to reach the deep system. This was facilitated often by backflow through precollectors and avalvular lymph capillaries in the dermis of the forearm, to reach eventually the few remaining lymph nodes in the axilla."
Conclusion: "Previously undetected lymph channels connecting the superficial and the deep lymphatic system had opened up because of the blockage of superficial lymphatic vessels caused by axillary dissection. It is presumed that these channels prevented lymphedema in this case."
Suami H et al., Changes in the Lymph Structure of the Upper Limb After Axillary Dissection: Radiogrpahic and Anatomoical Study in a Human Cadaver, Plast Reconstr Surg, 2007.
In the Suami H et al. 2007 study, the researchers' objective was to determine the differences in lymph structure in the upper arm following an axillary dissection.
To this end, the researchers compared a normal arm to an arm that had undergone an axillary dissection in the cadaver of an 81 year old woman..
Results: "...The mastectomy side showed remarkable differences and revealed that the lymph node clearance in the axilla had been incomplete on that side." As expected, there was an "obliteration of superficial lymph vessels". Also, there was "interval lymph node enlargement" in the arm. "The major difference was the almost complete absence of the superficial lymphatic network in the right arm, proximal to the elbow, because of fibrosis and blockage of the lymphatic channels. A circuitous pathway was identified that bypassed the blocked lymphatics in the arm to reach the deep system. This was facilitated often by backflow through precollectors and avalvular lymph capillaries in the dermis of the forearm, to reach eventually the few remaining lymph nodes in the axilla."
Conclusion: "Previously undetected lymph channels connecting the superficial and the deep lymphatic system had opened up because of the blockage of superficial lymphatic vessels caused by axillary dissection. It is presumed that these channels prevented lymphedema in this case."
Suami H et al., Changes in the Lymph Structure of the Upper Limb After Axillary Dissection: Radiogrpahic and Anatomoical Study in a Human Cadaver, Plast Reconstr Surg, 2007.
LYMPHEDEMA DEFINTION
"Lymphedema is caused by a reduction in lymph transport capacity associated with the cancer and its treatment (e.g., surgery, radiation therapy ,and chemotherapy), and results in the accumulation of protein-rich fluid in the interstitial space. Lymphedema results in disfigurement, discomfort, and functional impairments. In addition, LE may precipitate cellulitis and lymphangitis." (Miaskowski et al., 2013.)
Indeed, "Lymphedema can be debilitating, causing scarring, discomfort, disfigurement, difficulty in exercising, walking or other daily activities. Some patients are unable to wear their usual clothing or jewelry because of the increased weight and size of their affected limbs. There’s no cure for the condition – treatment generally centers on controlling pain and reducing swelling." (See https://www.ucsf.edu/news/2013/04/105101/genetic-markers-linked-development-lymphedema-breast-cancer-survivors.)
Indeed, "Lymphedema can be debilitating, causing scarring, discomfort, disfigurement, difficulty in exercising, walking or other daily activities. Some patients are unable to wear their usual clothing or jewelry because of the increased weight and size of their affected limbs. There’s no cure for the condition – treatment generally centers on controlling pain and reducing swelling." (See https://www.ucsf.edu/news/2013/04/105101/genetic-markers-linked-development-lymphedema-breast-cancer-survivors.)
ASSESSMENT OF LYMPHEDEMA - BIOELECTRICAL IMPEDANCE DEVICE
How to assess for lymphedema? Assessment of lymphedema has been very difficult in the past. But the advent of a single-frequency bioelectrical impedance device has made it more precise. This device "...measures the changes in extracellular fluid when compared with the unaffected contralateral upper extremity. A score increase of 10 or more or one that registers outside the normal range should prompt intervention with a compression garment or a referral for physical therapy. Some investigators report that measurement of bioimpedance has improved sensitivity in detecting subclinical lymphedema up to 4 months prior to standard measurement changes. For these reasons, the current National Lymphedema Network (NLN) breast cancer–related lymphedema guidelines recommend that all breast cancer patients receive pre- and post-treatment measurements (of any type) on both arms and encourage the use of bioimpedance spectroscopy or infrared perometry as alternatives to tape measures, to limit measurement variations."
( See Dr. Sarah McLaughlin, Lymphedema: Separating Fact From Fiction, Oncology, 2012.)
( See Dr. Sarah McLaughlin, Lymphedema: Separating Fact From Fiction, Oncology, 2012.)
WHO WILL GET LYMPHEDEMA?
MORE THAN TEN LYMPH NODES TAKEN IN A SENTINEL LYMPH NODE BIOPSY? NO LYMPHEDEMA! WHAT IF BETWEEN 10 AND 17 LYMPH NODES TAKEN IN AXILLARY DISSECTION? 11% LYMPHEDEMA!
IS IT THE "RELATIVE MAGNITUDE OF LYMPHATIC DESTRUCTION", RATHER THAN NUMBER OF NODES, THAT INCREASES THE RISK OF LYMPHEDEMA?
Dr. McLaughlin commented, "Perhaps it is the relative magnitude of lymphatic destruction and individual patient ability to form collateral lymphatic channels, rather than the number of nodes removed, that influences lymphedema risk."
YES! ... Lymphatic destruction, which does not necessarily mean the number of lymph nodes, taken, according to Dr. Sarah McLaughline's wonderful 2012 article.
Incidence of lymphedema? "Cancer-related lymphedema ranges from 0 to 3% after lumpectomy alone to as high as 65% to 70% after a modified radical mastectomy (removal of breast and axillary lymph nodes) with regional nodal radiation."
When? Within three years of treatment, 80% of the breast cancer patients who will ultimately get lymphedema have already been affected. "The risk persists years later as the remaining 10% to 20% will develop lymphedema at a rate of 1% per year." Since one study found that nearly 50% of 263 patients got lymphedema by 20 years, lymphedema is likely more common than realized.
Factors promoting lymphedema? Since a sentinel lymph node biopsy involves usually just one or more lymph nodes being taken, there are definitely significantly reduced rates of lymphedema following a sentinel node biopsy. 6 to 60 month trials found 0% to 7% of lymphedema in patients receiving sentinel node biopsies, and 12% to 16% rates in axillary lymph node biopsies (all nodes are taken)."These studies certainly support the concept that the risk of lymphedema is proportional to the extent of axillary surgery." In fact, one study "suggests that it is not the number of lymph nodes removed but instead the degree of dissection and disruption of the lymphatic system that results in lymphedema. The authors reviewed 600 women having sentinel lymph biopsy with a median follow-up of 5 years and found an overall incidence of lymphedema of 5%."
What is really interesting is that a subset of women who had more than 10 lymph nodes removed during a sentinel node biopsy did not develop lymphedema. To the contrary, women who had between 10-17 lymph nodes taken during axillary clearance had an 11% lymphemeda rate
A "complex" relationship exists between the number of nodes taken and the onset of lymphedema.
Dr. McLaughlin commented, "Perhaps it is the relative magnitude of lymphatic destruction and individual patient ability to form collateral lymphatic channels, rather than the number of nodes removed, that influences lymphedema risk. For example, a patient with many nodes removed at sentinel biopsy and no finding of lymphedema may have more lymphatic collaterals and therefore will have suffered relatively less lymphatic disruption despite a larger than 'normal' number of sentinel lymph nodes removed. On the other hand, women having axillary lymph node dissection and a relatively small number of total nodes excised may have suffered an overall greater degree of lymphatic disruption and therefore develop lymphedema. Unfortunately, the number of nodes within each patient’s nodal basin and the patient’s ability to protect or form new lymphatic collaterals during or after treatments is unknown. Therefore, simply the number of nodes removed may be insufficient to determine lymphedema risk."
Radiation? "If, however, the degree of lymphatic disruption or damage is the key driver of lymphedema risk, then it is plausible that radiation can also act primarily or synergistically to influence lymphedema risk. Axillary radiation alone is not without complications, as these patients can have a 2- to 4.5-fold increase in the risk of lymphedema. A recent meta-analysis ... finds lymphedema in 9% to 65% of patients after lumpectomy alone (no nodal surgery) and regional nodal radiation, and in 58% to 65% of women after mastectomy alone and regional nodal radiation. Additionally, the synergistic effect of surgery and radiation is well documented to result in a 3.5- to 10-fold higher risk of lymphedema when compared with surgery alone."
(See Dr. Sarah McLaughlin, Lymphedema: Separating Fact From Fiction, Oncology, 2012.)
WHO WILL GET LYMPHEDEMA? GENETIC PREDISPOSITION EXPLORED!
MIX OF BODY MASS INDEX, MORE ADVANCED BREAST CANCER, MORE LYMPH NODES TAKEN, AND GENE VARIATIONS?
"Patients with lymphedema had a significantly higher body mass index, more advanced disease, and a higher number of lymph nodes removed. Genetic associations were identified for four genes,,,"
Miaskowski C et al., 2014
Miaskowski C et al., 2014
In the Miaskowski C et al. 2013 study, the researchers sought to determine why some breast cancer patients develop lymphedema.
Their study evaluated over four hundred breast cancer patients, 155 of whom had lymphedema in the arm or fingers on the affected side. Testing included bioimpedance spectroscopy (which measures increases in the fluid in the arm) and DNA blood samples.
Results: "Patients with lymphedema had a significantly higher body mass index, more advanced disease, and a higher number of lymph nodes removed. Genetic associations were identified for four genes (i.e., lymphocyte cytosolic protein 2 (rs315721), neuropilin-2 (rs849530), protein tyrosine kinase (rs158689), vascular cell adhesion molecule 1 (rs3176861)) and three haplotypes (i.e., Forkhead box protein C2 (haplotype A03), neuropilin-2 (haplotype F03), vascular endothelial growth factor-C (haplotype B03)) involved in lymphangiogensis and angiogenesis."
From the UCSF Press Release: According to Bradley Aouizerat, PHD, "The genetic markers found in our study make perfect sense. These genes are 'turned on' later in the development of our lymph system and blood vessels. They appear to play a role in the ability of our lymphatic system to function on an ongoing basis. It is possible in some individuals who have changes in these genes, that lymphedema could develop after an injury like breast cancer surgery because these genes do not function properly."
Conclusion: "These genetic associations suggest a role for a number of lymphatic and angiogenic genes in the development of lymphedema following breast cancer treatment."
(For the study, see Miaskowski C et al., Lymphatic and Angiogenic Candidate Genes Predict the Development of Secondary Lymphedema Following Breast Cancer Surgery, PLOS One. 2013. ... For the UCSF Press Release, Elizabeth Fernandez, 2013,https://www.ucsf.edu/news/2013/04/105101/genetic-markers-linked-development-lymphedema-breast-cancer-survivors.)
Their study evaluated over four hundred breast cancer patients, 155 of whom had lymphedema in the arm or fingers on the affected side. Testing included bioimpedance spectroscopy (which measures increases in the fluid in the arm) and DNA blood samples.
Results: "Patients with lymphedema had a significantly higher body mass index, more advanced disease, and a higher number of lymph nodes removed. Genetic associations were identified for four genes (i.e., lymphocyte cytosolic protein 2 (rs315721), neuropilin-2 (rs849530), protein tyrosine kinase (rs158689), vascular cell adhesion molecule 1 (rs3176861)) and three haplotypes (i.e., Forkhead box protein C2 (haplotype A03), neuropilin-2 (haplotype F03), vascular endothelial growth factor-C (haplotype B03)) involved in lymphangiogensis and angiogenesis."
From the UCSF Press Release: According to Bradley Aouizerat, PHD, "The genetic markers found in our study make perfect sense. These genes are 'turned on' later in the development of our lymph system and blood vessels. They appear to play a role in the ability of our lymphatic system to function on an ongoing basis. It is possible in some individuals who have changes in these genes, that lymphedema could develop after an injury like breast cancer surgery because these genes do not function properly."
Conclusion: "These genetic associations suggest a role for a number of lymphatic and angiogenic genes in the development of lymphedema following breast cancer treatment."
(For the study, see Miaskowski C et al., Lymphatic and Angiogenic Candidate Genes Predict the Development of Secondary Lymphedema Following Breast Cancer Surgery, PLOS One. 2013. ... For the UCSF Press Release, Elizabeth Fernandez, 2013,https://www.ucsf.edu/news/2013/04/105101/genetic-markers-linked-development-lymphedema-breast-cancer-survivors.)
WHO WILL GET LYMPHEDEMA? - CYTOKINES EXPLORED - MIX OF BODY MASS INDEX, MORE ADVANCED BREAST CANCER MORE LYMPH NODES TAKEN, GENETIC LYMPHATIC VARIATIONS, AND PRO-AND ANTI-INFLAMMATORY CYTOKINE VARIATIONS?
As described above, the UCSF research team identified both phenotype -such as body mass index, number of lymph nodes taken, advanced stage of the disease, and sentinel node biopsy - as well as four discovered gene variants of lymphatic and angiogenesis pathways that increased the risk of lymphedema.
This UCSF group then continued their lymphedema research in a 2014 study, with an objective to find out if pro- and anti-inflammatory cytokines might be associated with lymphedema.
Results: "Genetic associations were identified for three genes (i.e., interleukin (IL4) 4 (rs2227284), IL 10 (rs1518111), and nuclear kappa factor beta 2 (NFKB2 (rs1056890)) associated with inflammatory responses..... For IL5, "One can hypothesize that dysregulation in the production of IL4 could lead to the development of soft tissue fibrosis and lymphatic dysfunction associated with lymphedema.... For IL10, "In addition to its anti-inflammatory effects, ... IL10 has anti-fibrotic properties and can inhibit excessive deposition of collagen and the transformation of fibroblasts to myofibroblasts." Note, too, that "polymorphisms in several candidate genes in IL10 and the IL10 receptor, which were not evaluated in this study, were associated with the development of lymphedema following infection with filarial parasites". Plus, "Patients who were homozygous (both copies mutated)for the rare allele in NFKB2 rs1056890 had a 3.1-fold increase in the odds of developing LE. NF-κB transcription factors play a role in diverse cellular processes including the regulation of angiogenesis, metastasis, cell proliferation, tumor promotion, suppression of apoptosis, and inflammation."
Conclusion: "These genetic associations suggest a role for a number of pro- and anti-inflammatory genes in the development of lymphedema following breast cancer treatment."
(See Leung G et al., Cytokine Candidate Genes Predict the Development of Secondary Lymphedema Following Breast Cancer Surgery, Lymphat Res Biol, 2014.)
2016 REVIEW STUDY - RESISTANCE EXERCISE TRAINING HELPFUL OR HARMFUL FOR BREAST CANCER SURVIVORS RELATIVE TO LYMPHEDEMA?
A Review Study of Literature: The objective was, "To determine if breast cancer survivors can perform resistance exercise training at sufficient intensities to elicit gains in strength without causing breast cancer flare up or incidence."
"The results of this review indicate that breast cancer survivors can perform resistance exercise training at high enough intensities to elicit strength gains without triggering changes to lymphedema There is strong evidence indicating that resistance training exercise produces significant gains in muscular strength without provoking breast cancer-related lymphedema."
Nelson NL, Breast Cancer-Related Lymphedema and Resistance Exercise: A Systematic Review, Strength Cond Res, 2016. (Also, go to the SELF-HELP EXERCISE section for more studies.)
A Review Study of Literature: The objective was, "To determine if breast cancer survivors can perform resistance exercise training at sufficient intensities to elicit gains in strength without causing breast cancer flare up or incidence."
"The results of this review indicate that breast cancer survivors can perform resistance exercise training at high enough intensities to elicit strength gains without triggering changes to lymphedema There is strong evidence indicating that resistance training exercise produces significant gains in muscular strength without provoking breast cancer-related lymphedema."
Nelson NL, Breast Cancer-Related Lymphedema and Resistance Exercise: A Systematic Review, Strength Cond Res, 2016. (Also, go to the SELF-HELP EXERCISE section for more studies.)
TWO DIFFERENT WAYS TUMOR CELLS CAN ENTER BODY: LYMPH FLUID AND BLOOD VESSELS
"Cancer cells that break away from
the primary tumor can invade the
circulation through either the blood vessels
or lymphatic vessels." Rizwan A et al., 2015.
the primary tumor can invade the
circulation through either the blood vessels
or lymphatic vessels." Rizwan A et al., 2015.
It is important to realize that, although this is a SURGERY-LYMPH NODES-LYMPHEDEMA section, tumor cells can enter the body either through the lymph vessels or the blood vessels.
When tumor cells escape from the tumor, the tumor cells either attach themselves to the outside wall of a lymph vessel or a blood vessel. "Then they must move through the vessel wall to flow with the blood or lymph to a new organ or lymph node."
Fortunately, most tumor cells that have escaped from the tumor die before they can start "growing somewhere" to form a metastasis. (See http://www.cancer.org/cancer/cancerbasics/lymph-nodes-and-cancer.)
More on how tumor cells spread to the blood vessels....
Previously the Harney group found "that breast cancer spreads when three specific cells are in direct contact: an endothelial cell (a type of cell that lines the blood vessels), a perivascular macrophage (a type of immune cell found near blood vessels), and a tumor cell that produces high levels of Mena, a protein that enhances a cancer cell's ability to invade. The site where these three cells come in direct and stable contact--called a tumor microenvironment of metastasis, or TMEM--is where tumor cells enter blood vessels." (See Blood vessel 'Doorway' Lets Breast Cancer Cells Spread Through Blood Stream, 2015,http://www.einstein.yu.edu/news/releases/1108/blood-vessel-doorway-lets-breast-cancer-cells-spread-through-blood-stream/.)
In their 2015 study, Harney A et al. extended their research and found via high-resolution two-photon microscopy that in primary mouse mammary cancer and in human xenografts in mice that "transient vascular permeability and tumor cell intravasation (flowing) occur simultaneously and exclusively at the tumor microenvironment (site) of metastasis." But what makes the blood vessels so permeable? The macrophages! The macrophages in this microenvironment release the growth factor, VEGF, which temporarily makes the blood vessels more permeable. So this is how the tumor cells can break away from the tumor and enter the blood stream to voyage to different sites and start metastases.
(See Harney A et al.,Real-Time Imaging Reveals Local, Transient Vascular Permeability, and Tumor Cell IntravasationStimulated by TIE2 Macrophage–Derived VEGFA, Cancer Discovery, 2015.)
It is important to realize that, although this is a SURGERY-LYMPH NODES-LYMPHEDEMA section, tumor cells can enter the body either through the lymph vessels or the blood vessels.
When tumor cells escape from the tumor, the tumor cells either attach themselves to the outside wall of a lymph vessel or a blood vessel. "Then they must move through the vessel wall to flow with the blood or lymph to a new organ or lymph node."
Fortunately, most tumor cells that have escaped from the tumor die before they can start "growing somewhere" to form a metastasis. (See http://www.cancer.org/cancer/cancerbasics/lymph-nodes-and-cancer.)
More on how tumor cells spread to the blood vessels....
Previously the Harney group found "that breast cancer spreads when three specific cells are in direct contact: an endothelial cell (a type of cell that lines the blood vessels), a perivascular macrophage (a type of immune cell found near blood vessels), and a tumor cell that produces high levels of Mena, a protein that enhances a cancer cell's ability to invade. The site where these three cells come in direct and stable contact--called a tumor microenvironment of metastasis, or TMEM--is where tumor cells enter blood vessels." (See Blood vessel 'Doorway' Lets Breast Cancer Cells Spread Through Blood Stream, 2015,http://www.einstein.yu.edu/news/releases/1108/blood-vessel-doorway-lets-breast-cancer-cells-spread-through-blood-stream/.)
In their 2015 study, Harney A et al. extended their research and found via high-resolution two-photon microscopy that in primary mouse mammary cancer and in human xenografts in mice that "transient vascular permeability and tumor cell intravasation (flowing) occur simultaneously and exclusively at the tumor microenvironment (site) of metastasis." But what makes the blood vessels so permeable? The macrophages! The macrophages in this microenvironment release the growth factor, VEGF, which temporarily makes the blood vessels more permeable. So this is how the tumor cells can break away from the tumor and enter the blood stream to voyage to different sites and start metastases.
(See Harney A et al.,Real-Time Imaging Reveals Local, Transient Vascular Permeability, and Tumor Cell IntravasationStimulated by TIE2 Macrophage–Derived VEGFA, Cancer Discovery, 2015.)