SURGERY
SURGERY AND ANESTHESIA
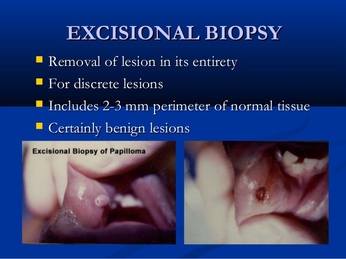
EXCISIONAL BIOPSY - REMOVAL OF THE ENTIRE TUMOR
SURGERY
SURGERY? NO SURGERY?
EDITORS' NOTE: WE DO NOT KNOW ANY ONE WHO SURVIVED BREAST CANCER WITHOUT REMOVING THE PRIMARY TUMOR.
NO SURGERY?
"There is mounting evidence that circulating tumor cells can move multi-dimensionally, seeding not just distant sites, but also tumors of origin." In a hormone-negative breast cancer model, genes could attract back cancer cells and facilitate entrance into the primary tumor. Dubbed, tumor "self-seeding". Comen and Norton, 2012.
YES, SURGERY? YOUNGER (Ages 40-49)?
In the Retsky M et al.2005 study, the researchers propounded the "mammogram paradox" theory that when younger women aged 40-49 have their tumors removed, the surgery "could remove the source of inhibitors of angiogenesis or growth factors could appear in response to surgical wounding".
HARMS WITH SURGERY?
More evidence in the Jesselsohn R et al. 2013 study below that biopsies prior to surgery create immune problems that harmfully spill over into the surgery period.
HARMS DUE TO PRIOR BIOPSY OR SURGERY ITSELF?
See the SURGERY-WINDOW OF OPPORTUNITY section to help ameliorate problems!
SURGERY? NO SURGERY?
EDITORS' NOTE: WE DO NOT KNOW ANY ONE WHO SURVIVED BREAST CANCER WITHOUT REMOVING THE PRIMARY TUMOR.
NO SURGERY?
"There is mounting evidence that circulating tumor cells can move multi-dimensionally, seeding not just distant sites, but also tumors of origin." In a hormone-negative breast cancer model, genes could attract back cancer cells and facilitate entrance into the primary tumor. Dubbed, tumor "self-seeding". Comen and Norton, 2012.
YES, SURGERY? YOUNGER (Ages 40-49)?
In the Retsky M et al.2005 study, the researchers propounded the "mammogram paradox" theory that when younger women aged 40-49 have their tumors removed, the surgery "could remove the source of inhibitors of angiogenesis or growth factors could appear in response to surgical wounding".
HARMS WITH SURGERY?
More evidence in the Jesselsohn R et al. 2013 study below that biopsies prior to surgery create immune problems that harmfully spill over into the surgery period.
HARMS DUE TO PRIOR BIOPSY OR SURGERY ITSELF?
See the SURGERY-WINDOW OF OPPORTUNITY section to help ameliorate problems!
NO SURGERY?
FEARFUL OF SPREADING BREAST CANCER CELLS? HOW ABOUT TUMOR "SELF-SEEDING"? TUMOR CELLS LEAVE TUMOR, RETURN?
TUMOR SELF-SEEDING
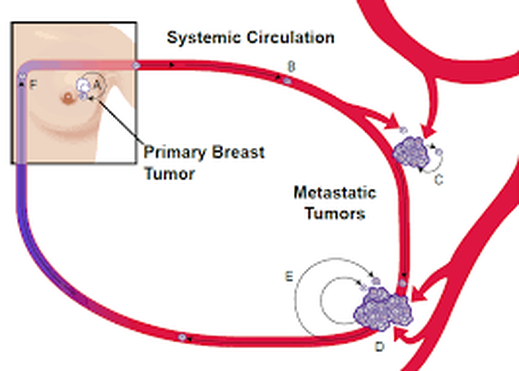
Cancer cells leaving a primary tumor can circle back to the primary tumor (A) or travel through the circulation, as a circulating tumor cell (B). Circulating tumor cells can then seed distant sites (C, D), seed metastatic tumor itself (E), or return back to the primary tumor site (F). (Elizabeth Comen and Larry Norton, 2012.)
Cancer cells leaving a primary tumor can circle back to the primary tumor (A) or travel through the circulation, as a circulating tumor cell (B). Circulating tumor cells can then seed distant sites (C, D), seed metastatic tumor itself (E), or return back to the primary tumor site (F). (Elizabeth Comen and Larry Norton, 2012.)
HAVE DENSE BREASTS?
TUMOR SEEDING AND DENSE BREASTS
"In the self-seeding model, a dense breast provides more scaffolding (collagen matrix) for self-seeds. Each self-seeding tumor, forming multiple masses, in turn promotes a more perilous framework for the surface/volume ratio of a transformed cellular state. ... Supporting this idea, recent research links increased stromal collagen to mammary tumor formation and metastasis." Comen and Norton, 2012.
TUMOR SEEDING AND DENSE BREASTS
"In the self-seeding model, a dense breast provides more scaffolding (collagen matrix) for self-seeds. Each self-seeding tumor, forming multiple masses, in turn promotes a more perilous framework for the surface/volume ratio of a transformed cellular state. ... Supporting this idea, recent research links increased stromal collagen to mammary tumor formation and metastasis." Comen and Norton, 2012.
"The self-seeding model challenges the unidirectional pattern of spread (of cancer cells)."
The tumor cells just don't go out, but they return!
".Born out of biological and clinical observations and buttressed by mathematical modeling, the self-seeding model accounts for the multidirectional flow between a primary tumor and metastatic sites. The self-seeding model posits that cancer cells are far more peripatetic, with the potential to seed regional and distant sites as well as return to the primary tumor."
(See Elizabeth Comen, Tracking the Seed and Tending the Soil: Evolving Concepts in Metastatic Breast Cancer, 2912.)
The tumor cells just don't go out, but they return!
".Born out of biological and clinical observations and buttressed by mathematical modeling, the self-seeding model accounts for the multidirectional flow between a primary tumor and metastatic sites. The self-seeding model posits that cancer cells are far more peripatetic, with the potential to seed regional and distant sites as well as return to the primary tumor."
(See Elizabeth Comen, Tracking the Seed and Tending the Soil: Evolving Concepts in Metastatic Breast Cancer, 2912.)
AFTER RADIATION THERAPY: "Dying cells in the breast may send out a signal that signals other cancer cells (circulating tumor cells) back to the side of the original tumor."
Krista Conger, 2014 See THERAPY-RADIATION section.
Krista Conger, 2014 See THERAPY-RADIATION section.
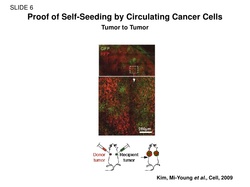
Adding to the self-seeding research.....
"Our observation that a mammary tumor can be seeded by circulating tumor cells derived from lung-metastatic nodules raises the possibility of reseeding after tumor excision as a potential cause of eventual local recurrence."
Kim M-Y et al., 2009
Kim M-Y et al., 2009
In the Kim M-Y et al. 2009 study, the researchers "postulated that circulating tumor cells may reinfiltrate an established tumor, enriching it with aggressive cells that have withstood a period of dissemination. This process, which we refer to as 'tumor self-seeding' could have consequences for tumor growth and the breeding of metastatic cell progenies".
In their mouse models of human cancer cells, which included human estrogen negative breast cancer cells, "...Tumor masses become readily seeded by circulating tumor cells derived from a separate tumor mass, from metastatic lesions, or from direct inoculation."
The shedding of tumor cells can occur in "large numbers and from early stages of tumor formation". But it is hard for these wandering cells to anchor themselves to another organ and become metastatic. Much easier it is for the cells to return to the tumor, as the "neovasculature of tumors is typically leaky".
How are circulating tumor cells able to return to the primary tumor?
"Self-seeding is actively driven by the ability of circulating tumor cells to sense attraction signals from the tumor and to extravasate (to force out into the fluid) in response to such signals." The best seeders are aggressive subpopulations of circulating tumor cells which have metastatic capabilities. For estrogen- negative breast cancer cells, "IL-6 and IL-8 act as circulating tumor cell attractants, whereas MMP1/collagenase-1 and the actin cytoskeleton component fascin-1 are mediatiors of circulating tumor cell infiltration into mammary tumors." Note that these are the same genes that are involved in metastasis for estrogen- negative breast cancer.
Larger tumors have always been associated with a poorer prognosis. It may not just "reflect the ability of larger cancers to release more cells of metastatic potential, but may in addition reflect the ability of such aggressive cells to self-seed, resulting in local-regional growth, acting in turn as a locus of expansion of those cells and priming for distant metastasis."
Especially interesting.... "Our observation that a mammary tumor can be seeded by circulating tumor cells derived from lung-metastatic nodules raises the possibility of reseeding after tumor excision as a potential cause of eventual local recurrence."
(See Kim M-Y et al., Tumor Self-Seeding by Circulating Cancer Cells, Cell, 2009.)
YES, SURGERY? - AGED 40-49? "MAMMOGRAM PARADOX"?
IN YOUNGER WOMEN, CAN SURGERY WAKE UP SLEEPING DISTANT MICROMETASTASES ACCELERATE TUMOR GROWTH?
In their mouse models of human cancer cells, which included human estrogen negative breast cancer cells, "...Tumor masses become readily seeded by circulating tumor cells derived from a separate tumor mass, from metastatic lesions, or from direct inoculation."
The shedding of tumor cells can occur in "large numbers and from early stages of tumor formation". But it is hard for these wandering cells to anchor themselves to another organ and become metastatic. Much easier it is for the cells to return to the tumor, as the "neovasculature of tumors is typically leaky".
How are circulating tumor cells able to return to the primary tumor?
"Self-seeding is actively driven by the ability of circulating tumor cells to sense attraction signals from the tumor and to extravasate (to force out into the fluid) in response to such signals." The best seeders are aggressive subpopulations of circulating tumor cells which have metastatic capabilities. For estrogen- negative breast cancer cells, "IL-6 and IL-8 act as circulating tumor cell attractants, whereas MMP1/collagenase-1 and the actin cytoskeleton component fascin-1 are mediatiors of circulating tumor cell infiltration into mammary tumors." Note that these are the same genes that are involved in metastasis for estrogen- negative breast cancer.
Larger tumors have always been associated with a poorer prognosis. It may not just "reflect the ability of larger cancers to release more cells of metastatic potential, but may in addition reflect the ability of such aggressive cells to self-seed, resulting in local-regional growth, acting in turn as a locus of expansion of those cells and priming for distant metastasis."
Especially interesting.... "Our observation that a mammary tumor can be seeded by circulating tumor cells derived from lung-metastatic nodules raises the possibility of reseeding after tumor excision as a potential cause of eventual local recurrence."
(See Kim M-Y et al., Tumor Self-Seeding by Circulating Cancer Cells, Cell, 2009.)
YES, SURGERY? - AGED 40-49? "MAMMOGRAM PARADOX"?
IN YOUNGER WOMEN, CAN SURGERY WAKE UP SLEEPING DISTANT MICROMETASTASES ACCELERATE TUMOR GROWTH?
The Retsky M et al. 2005 study posed the question, "What is causing apparently healthy young women (40-49) to die from breast cancer three years after the start of screening?"
Surgery!
The researchers, in studying what they dubbed the 'mammography paradox', "studied relapse patterns using a breast cancer database of 1173 pre- and postmenopausal, node-negative and -positive patients treated with surgery only and having 16–20 years of follow-up."
Relapse periods:
Early peak: An early relapse "occurs 8-10 months after surgery for young women with node-positive disease." Computer models "suggested that surgery probably instigated angiogenesis in dormant distant disease in approximately 20% of these cases.... . Calculations based on our data predict surgery-induced angiogenesis would accelerate disease by a median of two years and produce 0.11 early deaths per 1000 screened young women."
Ensuing peaks: " ... A sharp peak at 18 months, a nadir at 50 months, and a broad peak at 60 months with a long tail extending to 15–20 years. Patients with larger tumors more frequently relapse in the first peak while those with smaller tumors relapse equally in both peaks."
" In premenopausal patients with node-positive disease, 20% relapsed within the first 10 months following resection. That is a far higher percentage than for any other grouping. For comparison, in that first 10-month period, the relapse rate was five times higher for node-positive patients as node-negative patients. Also in that same period, the relapse rate was twice as high for premenopausal as postmenopausal patients. So the high frequency of relapse in the first 10 months after surgery was mainly peculiar to premenopausal node-positive patients."
Researchers' Observations: The researchers pondered whether, in the old days before all of this surgery, in breast cancer, there would just be dormant distant micrometastases, allowing the women to live an extra two years and have more children. ... Another observation: "... Our conclusions might provide a scientific basis for the often-debunked myth that 'cancer spreads when the air hits it'. The effect we describe would make it seem as though cancer spreads after surgery, while of course the cancer had already spread, but only escapes long-lasting pre-angiogenic dormancy as a biological sequel of surgery."
Conclusion: "Surgery-induced angiogenesis could account for the mammography paradox for women aged 40–49 and the bimodal relapse hazard pattern. According to the proposed biology, removing tumors could remove the source of inhibitors of angiogenesis or growth factors could appear in response to surgical wounding."
(See Retsky M et al., Does Surgery Induce Angiogenesis in Breast Cancer? Indirect Evidence From Relapse Pattern and Mammography Paradox, International Journal of Surgery, 2005.)
SURGERY - HARM
COMPARING BIOPSY AND SURGERY IMMUNE SYSTEM VARIATIONS
"CANCER BIOPSIES REVEAL ACTIVATION OF AN IMMUNE RESPONSE"!
As we learned in the Mathenge E et al. 2014 study in the SURGERY-BIOPSY section, there was imunosuppression following a core needle biopsy in a mouse model.
Enter the Jeselsohn R et al. 2013 study....
The Jeselsohn research team found that there were IMMUNE CHANGES between a core needle biopsy and an excisional biopsy to remove the entire tumor.
The researchers sought "to determine if there are variations in gene expression levels between core biopsies of early breast cancer specimens compared to the matched excisional biopsies."
The researchers found "that while the majority of the transcripts (immune) did not vary between the two biopsies, there was evidence of activation of immune related genes in response to the first biopsy."
Specifically, "Our study is notable for significant differences between core needle biopsies and excisional biopsy (surgery) in the expression level of 14 transcripts of which the majority are immune related. Of particular interest is the difference in CD68, a marker for tumor-associated macrophages. .In human breast tumors, infiltrating tumor-associated macrophages correlate with poor prognostic features, higher tumor grade and decreased disease free survival.. This may be, in part due to increased angiogenesis that has been shown in mouse models of mammary gland tumors and in human breast tumors where CD68 levels correlate with VEGF expression, .... The recruitment of CD68 positive tumor-associated macrophages after a core needle biopsy raises the concern that this recruitment may occur after each breast cancer biopsy. This raises the question of whether core needle biopsy may have a deleterious effect on outcome through the stimulation of the recruitment of tumor-associated biopsies and this is of particular concern as many neoadjuvant clinical trials include additional biopsies."
Conclusion: "There was evidence of activation of immune related genes in response to the first biopsy, and further investigations of the immune changes after a biopsy in early breast cancer seem warranted."
(EDITORS' NOTE): The researchers are calling for "window of opportunity" studies to try to mitigate the core needle biopsy's damage to the immune system. While the Editors are big proponents of ONLY having an excisional biopsy - all of the tumor to be removed at once - if a person decides to do surgery, all surgeries provide huge, open windows of opportunity to help lessen the inflammation, etc. SEE SURGERY-WINDOW OF OPPORTUNITY section.)
(For the study, see Jeselsohn, R et al., Digital Quantification of Gene Expression in Sequential Breast Cancer Biopsies Reveals Activation of an Immune Response, PLOS One, 2013.)
HARMS WITH SURGERY?
To recapitulate, the Jesselsohn R et al. 2013 study cited above illustrates that biopsies prior to surgery create immune problems that can harmfully spill over into the surgery period.
To recapitulate, the Jesselsohn R et al. 2013 study cited above illustrates that biopsies prior to surgery create immune problems that can harmfully spill over into the surgery period.
SURGERY - HARM
NEUTROPHIL-LYMPHOCYTE RATIO (INFLAMMATION) AFTER SURGERY INFLAMMATION A SIGNIFICANT MARKER FOR BREAST CANCER OUTLOOK
In the Lee SK et al. 2015 study, the researchers "investigated the prognostic significance of perioperative inflammation with the neutrophil-lymphocyte ratio in breast cancer".
What is the neutrophil-lymphocyte ratio? Neutrophils are the immune system's first line of defense, whereas the lymphocytes help to regulate the inflammation.
"Inflammation is associated with worse outcomes in cancer. Surgeries induce an acute inflammatory response and could impact the clinical outcomes in breast cancer." And in this retrospective study, the surgical records of 3,116 breast cancer patients were reviewed as to the neutrophil-lymphocyte ratio one week before surgery and one week after surgery.
Results: "...The neutrophil-lymphocyte ratio in postoperative week 1, total mastectomy, the presence of lymphovascular invasion, a higher nuclear grade and pathologic TNM stage, and negative hormone receptor and subtypes were factors associated with poor disease-specific survival. The neutrophil-lymphocyte ratio in postoperative week 1 remained a significant prognostic factor ... A cutoff level of 5.2, determined by the minimum p value approach, was found to be a significant level for discriminating the impact on breast cancer-specific mortality."
Conclusion: "Immediate postoperative inflammation is an important prognostic marker in breast cancer patients."
(See Lee SK et al., Immediate Postoperative Inflammation is an Important Prognostic Factor in Breast Cancer, Oncology, 2015.)
"Why Inflammation may
be harmful ...
On to inflammation's
activation
of STAT
after surgery ....."
SURGERY - HARM
SURGERY-INDUCED INFLAMMATION MAY ENGAGE STAT3 PATHWAY, LEADING TO STEM-LIKE FEATURES.
__________________________________________________________
"Surgery represents an event that acutely causes both inflammation and wound healing response, strongly suggesting that it may represent a perturbing factor in the process of local recurrence or metastasis development in humans."
"Wound fluids in breast cancer patients after surgery contain factors that are highly stimulatory of the self renewal and stemlike phenotypes of breast cancer cells."
______________________________________________________
In the Segatto I et al. 2014 study, the researchers found that wound fluids following breast cancer surgery cause a cascade of inflammatory reactions that activate STAT.
What is STAT? "STAT (Signal Transducer and Activator of Transcription) regulates many aspects of growth, survival, and differentiation in cells." (See Wikipedia.org.)
The researchers discovered that, ... "Wound fluids induced the enrichment of breast cancer cells with stem-like phenotypes, via activation of STAT3. ... Stem-like phenotype of cancer cells may account for their capability to disease relapse, even when the primary tumor is eradicated. These tumor-initiating cells share properties of self renewal and differentiation with normal stem cell counterparts, although in tumor-initiating cells, these processes are deregulated."
Specifically, "In vitro, wound fluids highly stimulated mammosphere formation and self-renewal of breast cancer cells. In vivo (a mouse model), STAT3 signaling was critical for breast cancer cell tumorigenicity and for the formation of local relapse after surgery."
Discussion: "Although the molecular mechanisms linking inflammation and cancer have long remained elusive, a role for inflammation in tumorigenesis is generally accepted. Also the process of wound healing to replace damaged tissue has been linked to cancer. Wound healing is a complex response that includes inflammation, neovascularization, matrix deposition and re-organization and involves a small population of replenishing stem cells giving rise to differentiated progeny that replace the damaged tissue. This characteristic is shared with the stem-like component of a growing tumor and in fact, epithelial tumors have been described as ‘wounds that do not heal’ because of the molecular and cellular similarities between features associated with wounds and carcinomas. Surgery represents an event that acutely causes both inflammation and wound healing response, strongly suggesting that it may represent a perturbing factor in the process of local recurrence or metastasis development in humans."
Conclusion: "Our findings highlight that the inflammatory response induced by surgery may engage STAT3 pathway activation in residual breast caner cells and this, in turn, leads to the acquisition of stem-like features."
(See Segatto I et al., Surgery-Induced Wound Response Promotes Stem-Like and Tumor-Initiating Features of Breast Cancer Cells, Via Stat3 Signaling, Oncotarget, 2014.)
SURGERY - ANESTHESIA
ANESTHESIA
QUOTES
"The problem is that the natural killer function is impaired by the surgical stress response by volatile anesthetics and by opioids. Regional analgesia reduces the surgical stress response and decreases the need for volatile anesthetics and opioids." Dr. Daniel Sessler, 2014.
"Our main finding is that apoptosis of the human breast carcinoma cell line MDA-MG-231 (ER- Negative) was significantly reduced when cells were treated with postoperative serum sevoflurane group compared with the propofol-opioid group." Jaura A et al., 2014.
"Our findings bring increased evidence to the hypothesis that paravertebral blocks through attenuation of surgical stress and reduced opioid consumption reduces the risk of developing metastasis." Oppfeldt A et al., 2013.
QUOTES
"The problem is that the natural killer function is impaired by the surgical stress response by volatile anesthetics and by opioids. Regional analgesia reduces the surgical stress response and decreases the need for volatile anesthetics and opioids." Dr. Daniel Sessler, 2014.
"Our main finding is that apoptosis of the human breast carcinoma cell line MDA-MG-231 (ER- Negative) was significantly reduced when cells were treated with postoperative serum sevoflurane group compared with the propofol-opioid group." Jaura A et al., 2014.
"Our findings bring increased evidence to the hypothesis that paravertebral blocks through attenuation of surgical stress and reduced opioid consumption reduces the risk of developing metastasis." Oppfeldt A et al., 2013.
SURGERY- ANESTHESIA
SOME ANESTHESIAS MAY BE MORE BENEFICIAL IN POSTOPERATIVE CANCER PROGNOSIS
BACKGROUND
"Surgery causes metabolic, neuroendocrine, inflammatory, and immunological stress and determines upregulation of major malignant molecular pathways involved in tumorigenesis The results of studies in vitro and in vivo show that the body’s response to surgical stress increases the likelihood of metastatic spread of cancer. Surgery also increases chances of tumor growth and metastasis, due to release of circulating cancer cells during surgical resection of cancer and to the inability of the immune system to neutralize them.
The role of anesthetics and analgesics in postoperative cancer recurrence has also been investigated. Although guidelines for appropriate use of anesthetics in surgical oncology have not yet been codified, these drugs are thought to affect the evolution of surgically treated tumors."
(See Fodale V et al., Anesthetic Techniques and Cancer Recurrence After Surgery, The Scientific World Journal, 2014.)
SOME DEFINITIONS OF ANESTHESIA:
"Sevoflurane ... is a sweet-smelling, nonflammable, highly fluorinated methyl isopropyl ether used as an inhalational anestheric for induction and maintenance of general anesthesia." (wikipedia.org.)
Paravertebral blocks are regional anesthesia blocks.
Opioids are "any chemicals, such as morphine, that resemble opiates in their pharmacological effects. The painkiller effects of opioids are due to decreased perception of pain, decreased reaction to pain, as well as increased pain tolerance". (wikipedia.org.)
" Propofol ... is a short-acting, intravenously administered hypnotic/amnestic agent. It uses include the induction and maintenance of general anesthesia, sedation for mechanically ventilated adults, and procedural sedation." (wikipedia.org)
SURGERY - ANESTHESIA
WHICH ANESTHESIA TECHNIQUE BETTER HELPS NATURAL KILLER CELLS AGAINST BREAST CANCER CELLS?
PROPOFOL-PARAVERTEBRAL BLOCK OR SEVOFLURANE-OPIOIDS?
Propofol-paravertebral block!...... According to the Buckley A et al. 2014 study!
In the Buckley A et al. pilot study, the researchers sought to determine which anesthetic technique would better promote NK (natural killer cell) activity against breast cancer cells. Note that, "Natural Killer T cells are a critical component of the anti-tumor immune response."
In this experiment, ten women donated serum before their breast cancer surgeries, then 24 hours after the surgery. In vitro, the serum was studied for natural killer cell activity against hormone-positive breast cancer cells.
Results: "An increase in NK cell CD107a [23 (2)% to 37(3)%] and apoptosis of the breast cancer cells [11 (1)% to 21 (2)%0] was observed with propofol-paravertebral block serum, but not sevoflurane-opioids serum, treated NK cells."
Conclusion: "Serum from subjects receiving propofol-paravertebral block resulted in greater NK cell cytotoxicity than serum from subjects receiving sevoflurane-opioids."
______________________________
EDITORS' NOTE: In October 2014 Anesthesiology News, Dr.Daniel Sessler stated, "The primary defense against new cancers and cancer recurrence is natural killer cells. The problem is that natural killer cell function is impaired by the surgical stress response by volatile anesthetics and by opioids. Regional analgesia reduces the surgical stress response and decreases the need for volatile anesthetics and opioids. Intravenous lidocaine is anti-inflammatory and appears to enhance natural killer cell function, and the same is true for COX-2 inhibitors. In theory, then, either or both may be helpful."
__________________________________________
Also, see the discussion of the analgesia KETOROLAC in the SURGERY - WINDOW OF OPPORTUNITY section.
(For the study, Buckley A et al., Effect of Anesthetic Technique on the Natural Killer Cell and Anti-Tumor Activity of Serum From Women Undergoing Breast Cancer Surgery: A Pilot Study, British Journal of Anesthesia, 2014 .. For Anesthesiology News, seehttp://www.anesthesiologynews.com/ViewArticle.aspx?d=Policy+&+Management&d_id=3&i=October+2014&i_id=1107&a_id=28364.}
SURGERY - ANESTHESIA
DOES A PARAVERTEBRAL NERVE BLOCK COMBINED WITH GENERAL ANESTHESIA DURING BREAST CANCER SURGERY HELP REDUCE BREAST CANCER RECURRENCE?
JUST MAYBE COULD THE PARAVERTEBRAL BLOCK BE INCREASING NK CELL ACTIVITY VITAL TO IMMUNE SYSTEM?
YES AND YES! ...... According to the Oppfeldt A et al. 2013 Abstract Presentation.
In the Oppfeldt A et al. 2013 Abstract Presentation, the researchers sought to determine "if paravertebral blocks combined with general anesthesia reduce the risk of breast cancer recurrence when compared to general anesthesia alone."
Excerpts from Science Daily: "Breast cancer patients who received the combination of a nerve block with general anesthesia for their breast cancer surgery had less cancer recurrence and were three times less likely to die than those who received only general anesthesia. Additionally, patients who received the nerve block needed less opioid pain relief from drugs such as fentanyl and oxycodone."
In this study, breast cancer patients "were divided into two groups, both having general anesthesia. In addition, the first group received a regional nerve block via four to six injections of local anesthetic. The second group received injections of saline. After six years, these patients' medical records were reviewed for death or cancer recurrence."
Results: "The study found that the death rate was significantly lower in the group who received the nerve block with general anesthesia. Ten percent of patients who received the nerve block and general anesthesia died compared to 32 percent of patients who received the placebo. Additionally, the rate of cancer recurrence was significantly less for the nerve block and general anesthesia group as compared to the placebo group: 13 percent had a recurrence while 37 percent of the placebo group's cancer returned. The nerve block and general anesthesia group also took significantly fewer opioids than the placebo group: an average of 45 mg of morphine compared to the patients with cancer recurrence who took an average of 58 mg of morphine."
Regional anesthesia and NK Cells? Here is the connection!
Excerpts from Medscape:
"Anesthesiologists should use regional anesthesia as much as possible during cancer surgery, because it seems to improve survival and cancer recurrence and is without the potential side effects of morphine," senior researcher Palle Carlsson, MD, DMSc, associate professor in the Department of Anesthesiology at Aarhus University Hospital in Denmark
And NK cells? "It is possible that paravertebral block improves outcomes by reducing surgical stress and increasing the number of natural killer cells, which are vital to the immune system and the body's defense against cancer, Dr. Carlsson explained. However, the women with better outcomes also had less postoperative use of opioids, and opioids are known to decrease natural killer cells, he added..
Conclusion from study: "Our findings bring increased evidence to the hypothesis that paravertebral blocks through attenuation of surgical stress and reduced opioid consumption reduces the risk of developing metastasis."
(For the study, see Oppfeldt a et al., American Society of Anesthesiologists (ASA) 2013 Annual Meeting: Abstract 4253. Presented October 15, 2013. For Science Daily, see "http://www.sciencedaily.com/releases/2013/10/131015191057.htm. For Medscape, see
http://www.medscape.com/viewarticle/812680#vp_1lh.}
SURGERY - ANESTHESIA
WHICH ANESTHESIA TECHNIQUE IS BETTER FOR KILLING CANCER CELLS (APOPTOSIS) - - PROPOFOL-VERTERBRAL BLOCK OR SEVOFLURANCE-OPIOIDS?
------------------------------------------------
Jura A et al., 2014 Study Editor's Key Points:
- "Some data suggest that anaesthetic technique might affect cancer outcome.
- Serum was obtained from a subset of patients enrolled in a larger clinical trial who were randomized to receive propofol-paravertebral or sevoflurane-opioid based anaesthesia.
- Postoperative serum from the sevoflurane anaesthesia group reduced cancer cell apoptosis in vitro. These preliminary findings suggest that anaesthetic technique might affect cancer cell metastatic potential during cancer surgery."
Propofol-vertebral block! .... According to the Jaura A et al. 2014 study, the propofol-vertebral block is better at increasing the apoptosis rate!
Apoptosis is the killing of cancer cells. If apoptosis isn't functioning properly, cancer may develop and progress. And anesthesia may have an effect on apoptosis.
The researchers tested anesthetics on the serum of women with a more aggressive breast cancer type, estrogen receptor-negative breast cancer. ER-negative women were given "either propofol general anesthesia along with a paravertebral analgesia, or the standard sevoflurance general anesthesia with opioid analgesia". Blood was taken immediately before the anesthesia and at one hour after the breast cancer surgery and tested on the estrogen receptor-negative cells for apoptosis rates.
.Background: Among other things, surgery can release cancer cells, reduce anti-angiogenesis, and induce stress which can inhibit NK cell activity. Enter anesthetics. "Anesthesia per se might also promote metastasis.... Impairing neutrophil, macrophage, T-cell, and NK cell function. Opioids are known to inhibit human cellular and humor al immune function ... Lower doses, typical of those used perioperatively, appear to be tumor-promoting. Postoperative pain suppresses cell-mediated immunity and enhances the tumor-promoting effects of surgery, suggesting that optimum perioperative analgesia might enhance metastasis resistance in cancer patients having surgery." In the past, these researchers had found that the propofol-paravertebral block, compared with sevoflurane, "decreases serum concentrations of pro-tumorigenic cytokines and matrix metalloproteinases (IL-1B, MMP-3, and MMP-9) and increases serum concentration of the anti-tumorigenic cytokine IL-10." Elsewhere, there was a finding that the propofol-paravertebral block is associated with reduced VEGF (induces angiogenesis}.
In this study, "Greater cancer cell apoptosis produced by postoperative serum from the propofol-paravertebral block group compared with the sevoflurane-opioid group could be attributable to an alteration in the molecular profile of the serum as a result of anesthetic technique." Indeed,"It is unlikely that sevoflurane or opioids have a persistent effect at 1 hour that is directly causing the difference in apoptosis we observed. Rather, it seems more plausible that these agents of those used in the propofol-paravertebral block group induce changes in serum that indirectly affect cancer cells or immune cells."
Conclusion: "These perliminary results suggest that anesthetic technique might affect the serum milieu in breast cancer patients to influence metastasis potential." The propofol-paravertebral block is better for apoptosis - killing cancer cells! (See Jaura A et al., Differential effects of Serum from Patients Administered Distinct Anaesthetic Techniques on Apoptosis in Breast Cancer Cells In Vitro: A Pilot Study, British Journal of Anesthesiology, 2014.)